-
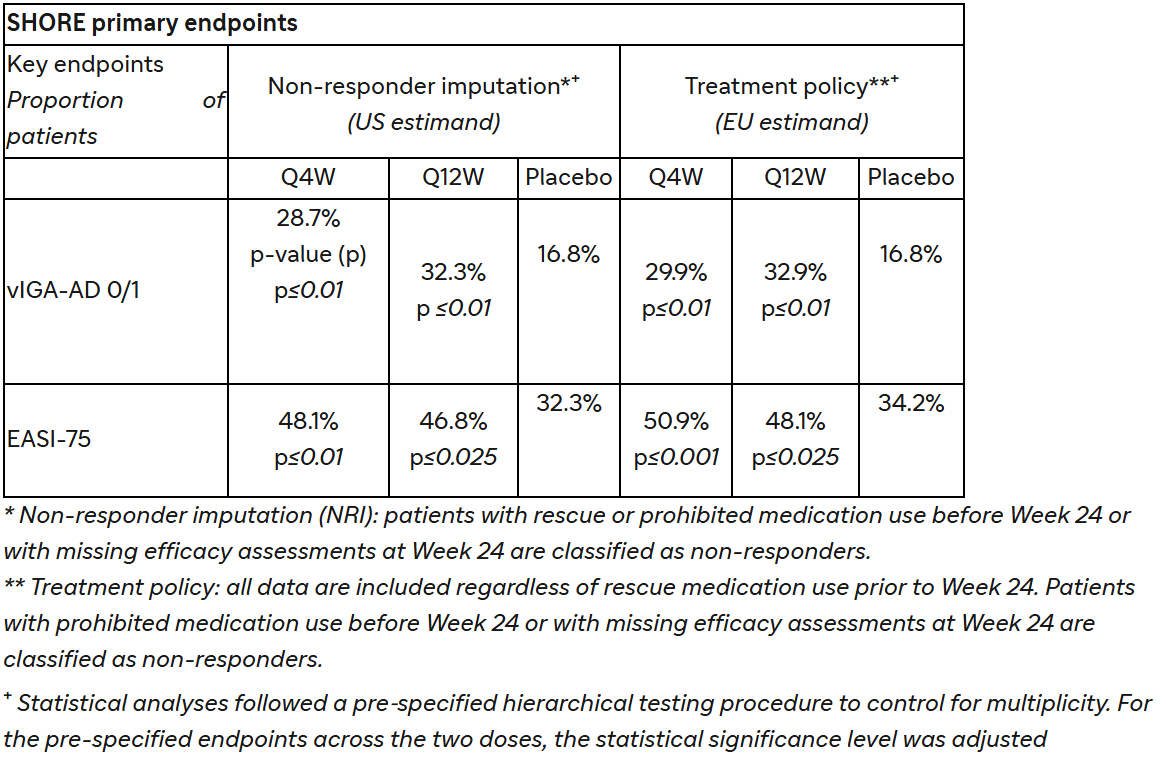
In the SHORE phase 3 study, amlitelimab in combination with topical therapies met all primary and key secondary endpoints at Week 24 with efficacy progressively increasing throughout the treatment period with some patients improving as early as Week 2
-
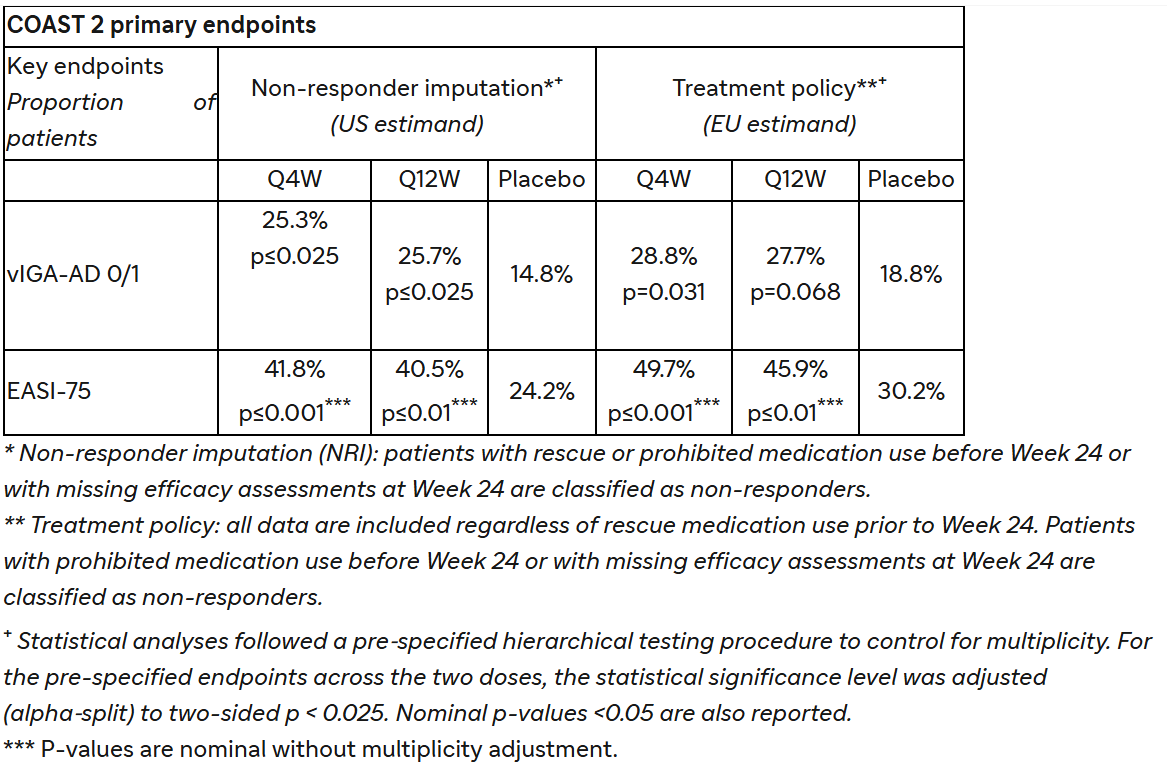
In the COAST 2 phase 3 study, amlitelimab demonstrated statistically significant efficacy on vIGA-AD 0/1, the primary endpoint as assessed for the US and US reference countries, and confirmed COAST 1 potential for Q12W dosing from the start; amlitelimab did not achieve statistical significance for the co-primary endpoints as assessed for the EU and EU reference countries
-
Based on the totality of data, Sanofi will move forward with global regulatory submissions for amlitelimab
-
A preliminary analysis of the ATLANTIS phase 2 study showed continued and progressive improvements with no evidence of plateau through Week 52, demonstrating the potential of OX40-ligand as an important new mechanism in AD
Paris, January 23, 2026. Following the positive COAST 1 (clinical study identifier: NCT06130566) results in September 2025, two additional global phase 3 studies – SHORE (clinical study identifier: NCT06224348) and COAST 2 (clinical study identifier: NCT06181435) – of amlitelimab, a fully human non-T cell depleting monoclonal antibody that selectively targets OX40-ligand (OX40L), today delivered a robust body of evidence that supports amlitelimab’s potential in the treatment of patients 12 years and older with moderate-to-severe atopic dermatitis (AD). In these two phase 3 studies amlitelimab was well-tolerated and the safety profile was consistent with previously reported data.
"Importantly, these results validate amlitelimab’s novel mechanism of action to block OX40-ligand without T-cell depletion and its promise to normalize the immune system over time," said Houman Ashrafian, Executive Vice President, Head of Research & Development at Sanofi. "The totality of data seen to date reinforce our confidence in amlitelimab’s potential to deliver both Q12W dosing from the start and progressive efficacy through Week 52. We look forward to sharing additional results, including longer-term data, as we move toward global regulatory submissions.”
For both SHORE and COAST 2 phase 3 studies, key endpoints were measured at Week 24 in patients aged 12 years and older with moderate-to-severe AD who received amlitelimab either every four weeks (Q4W) or every 12 weeks (Q12W). For US and US reference countries, the single primary endpoint was the proportion of patients with a validated investigator global assessment scale for AD (vIGA-AD) of 0 (clear) or 1 (almost clear) and a reduction from baseline score of ≥2 points, analyzed using non-responder imputation (US estimand). For the EU, EU reference countries and Japan, the co-primary endpoints comprised the proportion of patients with vIGA-AD 0/1 and a reduction from baseline score of ≥2 points along with the proportion of patients reaching a 75% or greater improvement in the eczema area and severity index total score (EASI-75), both analyzed using treatment policy (EU estimand).
SHORE study
In the SHORE study, amlitelimab, dosed either Q4W or Q12W in conjunction with medium-potency background topical corticosteroids (TCS) with or without topical calcineurin inhibitors (TCI), met all primary and key secondary endpoints compared to placebo plus TCS with or without TCI at Week 24, across both US and EU estimands.
COAST 2 study
In the COAST 2 study, amlitelimab monotherapy dosed either Q4W or Q12W met the primary endpoint of the proportion of patients achieving vIGA-AD 0/1 and a reduction from baseline score of ≥2 points compared to placebo at Week 24, as assessed for the US and US reference countries. The key secondary endpoint of the proportion of patients who achieved vIGA-AD 0/1 with barely perceptible erythema (BPE), as assessed for the US and US reference countries, did not achieve statistical significance. For the EU and EU reference countries, amlitelimab dosed either Q4W or Q12W did not achieve statistical significance for the co-primary endpoints of proportion of patients achieving vIGA-AD 0/1 and EASI-75 compared to placebo. Therefore, in accordance with hierarchical statistical testing procedures, p-values presented below for additional secondary endpoints reflect nominal significance without adjustment for multiplicity.
The most common treatment-emergent adverse events (TEAEs) in SHORE (≥5% in any dose arm; pooled amlitelimab vs. placebo) were nasopharyngitis (9.5% vs 12.5%), upper respiratory tract infection (7.9% vs 4.4%), dermatitis atopic (2.7% vs 5.6%). Overall, rates of TEAEs, serious adverse events, and TEAEs resulting in treatment discontinuation were similar in the pooled amlitelimab arms and placebo arm.
The most common TEAEs in COAST 2 (≥5% in any dose arm; pooled amlitelimab vs. placebo) were nasopharyngitis (5.9% vs 7.4%), upper respiratory tract infection (4.8% vs 4.0%), dermatitis atopic (5.3% vs 2.7%). Overall, rates of TEAEs, serious adverse events, and TEAEs resulting in treatment discontinuation were similar in the pooled amlitelimab and placebo arms.
About the SHORE study
SHORE was a randomized, double-blind, placebo-controlled, parallel-group, 3-arm, multinational, multicenter phase 3 study to evaluate the efficacy and safety of amlitelimab in combination TCS with or without TCI in 596 participants aged 12 years and older with moderate-to-severe AD. Key objectives include measuring the efficacy and safety of amlitelimab compared to placebo at Week 24 when used in combination with TCS/TCI. In the study, amlitelimab was administered at a dose of 250 mg (125 mg for those with body weight <40 kg) on either a Q4W or Q12W schedule following a loading dose of 500 mg (250 mg for those with body weight <40 kg). Patients were given medium-potency TCS/TCI, applied up to twice daily to treat active lesions, and were instructed to reduce the dose to three times weekly or discontinue use based on lesion control or clearance. The study included sites across the North America, EU, Argentina, Chile, Brazil, Turkey, Canada, China, and Japan.
About the COAST 2 study
COAST 2 was a randomized, double-blind, placebo-controlled, parallel-group, 3-arm, global, multicenter phase 3 study to evaluate the efficacy and safety of amlitelimab monotherapy in 547 adults and adolescents aged 12 years and older with moderate-to-severe AD. Key objectives included measuring the efficacy and safety of amlitelimab compared to placebo at Week 24. In the study, amlitelimab was administered at a dose of 250 mg (125 mg for those with body weight <40 kg) on either a Q4W or Q12W schedule following a loading dose of 500 mg (250 mg for those with body weight <40 kg). The study included sites across the US, EU, United Kingdom, Argentina, Chile, Mexico, South Africa, Turkey, China, and Japan.
About the ATLANTIS study
ATLANTIS is a phase 2 global, single-arm, open-label, long-term safety study of amlitelimab for the treatment of patients 12 years or older with moderate to severe atopic dermatitis (AD) with a duration of up to 268 weeks. In the study, patients received weight-based amlitelimab Q4W (250 mg [125 mg for those with body weight <40 kg] following a loading dose of 500 mg [250 mg for those with body weight <40 kg]). The primary outcome measures of the ATLANTIS study are the percentage of participants who experienced TEAEs and TESAEs from baseline. Efficacy outcome measures included the proportion of patients with a vIGA-AD 0/1 and the proportion of participants achieving EASI-75. The study has enrolled 963 patients to date and includes sites across North America, Latin America, EU, United Kingdom, Turkey, South Africa, India, Australia, South Korea, Taiwan, China, and Japan.
About amlitelimab
Amlitelimab (SAR445229, KY1005) is a fully human, non-T cell depleting monoclonal antibody that blocks the OX40L, a key immune regulator. With its novel mechanism of action, amlitelimab selectively blocks OX40L signaling during the inflammatory prequel, the initiating phase of an overactive immune system, to potentially normalize T-cell-mediated inflammation without T-cell depletion.
About Sanofi
Sanofi is an R&D driven, AI-powered biopharma company committed to improving people’s lives and delivering compelling growth. We apply our deep understanding of the immune system to invent medicines and vaccines that treat and protect millions of people around the world, with an innovative pipeline that could benefit millions more. Our team is guided by one purpose: we chase the miracles of science to improve people’s lives; this inspires us to drive progress and deliver positive impact for our people and the communities we serve, by addressing the most urgent healthcare, environmental, and societal challenges of our time.