- Trial met primary efficacy endpoint at week 12
- EVO301 produced rapid, statistically significant EASI reductions at weeks 4, 8 and 12 versus placebo
- 33% placebo-adjusted improvement in EASI at week 12
- 23% of EVO301 patients achieved IGA 0/1 at week 12
- Company to hold conference call and webcast at 8:30 a.m. ET today
February 10, 2026--PALO ALTO, Calif. & NEW YORK--(BUSINESS WIRE)-- Evommune, Inc. (NYSE: EVMN) (the “Company” or “Evommune”), a clinical-stage biotechnology company developing innovative therapies that target key drivers of chronic inflammatory diseases, today announced positive top-line results from its randomized, double-blind, placebo-controlled Phase 2a trial evaluating EVO301. This long-acting fusion protein consisting of an interleukin-18 (IL-18) binding protein and an anti-serum albumin Fab-associated domain, achieved highly statistically significant outcomes in adult patients with moderate-to-severe atopic dermatitis (AD). The 70-patient trial was designed to evaluate the safety and efficacy of intravenous dosing of 5mg/kg on day 1 and day 28 (n=48 active, n=22 placebo) over 12 weeks.
“These data underscore that impacting pathways beyond only Th2 biology can meaningfully contribute to AD disease activity. EVO301’s ability to target the novel IL-18 mechanism and show clinically relevant treatment activity, without side effects, could offer real benefit for patients in such a heterogeneous disease,” said Dr. Mark G. Lebwohl, Dean for Clinical Therapeutics and Chairman Emeritus of the Department of Dermatology at the Icahn School of Medicine at Mount Sinai. “There is an urgent need for new treatment options for the growing number of patients suffering from AD. With rapid onset and durable responses, as evidenced by meeting the primary endpoint at weeks 4, 8 and 12 after only two doses, this trial supports the potential of EVO301 to become a front-line biologic treatment in AD, if approved.”
“This is a big milestone for Evommune and we are pleased to report unequivocally positive data that validate IL-18 inhibition and show significant activity of EVO301 in moderate-to-severe AD patients. These data support our plans to move EVO301 into a Phase 2b dose-ranging trial where we can optimize the dose and seek to further improve patient outcomes," said Luis Peña, President and Chief Executive Officer at Evommune. “With multiple programs now having positive Phase 2 data, we are poised to grow into a significant company in the immunology space,” added Mr. Peña.
Key data highlights include:
-
Primary Endpoint: The trial met its primary endpoint; a Bayesian success criterion related to the difference between active and placebo in the percent improvement in baseline in the Eczema Area and Severity Index (EASI). While the success criterion required at least 75% of the posterior distribution to be an improvement of at least 8% over placebo, the results of the study demonstrated 99.8% of the posterior distribution met that threshold. Furthermore, when analyzed by the more commonly used frequentist method, statistical significance was achieved at weeks 4, 8 and 12 at p<0.01.
-
% Reduction in EASI at weeks 4, 8 and 12:
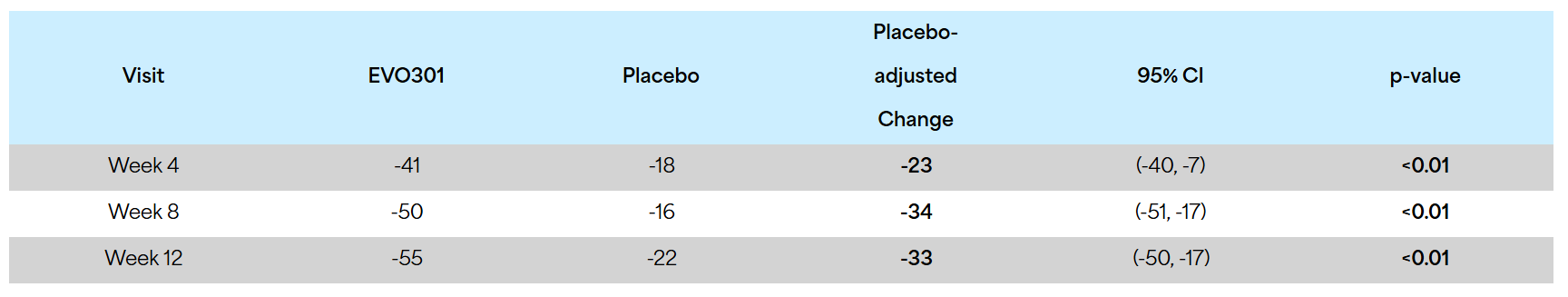
Additionally, 23% of patients treated with EVO301 (vs 0% placebo) achieved vIGA-AD 0/1 (percent of patients achieving a score of 0 or 1 on the validated Investigator’s Global Assessment for Atopic Dermatitis with ≥ 2-point reduction from baseline) at week 12.
-
Pharmacokinetics (“PK”): Consistent with the Phase 1 healthy volunteer trial; PK and target engagement data continue to support a Q4 week dosing regimen.
-
Safety Profile: EVO301 was well tolerated, with no related serious or severe adverse events reported, no treatment related discontinuations due to adverse events and no meaningful differences in events between the active and placebo groups.
-
Biomarkers: Robust reduction of both Th2 and non Th2 inflammatory biomarkers in AD including CCL-17 (TARC), CCL-22 and IL-22.
Next Steps
Full results from the Phase 2a trial will be presented at a future scientific conference. The Company continues to advance planning for a Phase 2b dose‑ranging trial in patients with AD, using a subcutaneous formulation of EVO301. The Company is also evaluating potential additional indications for EVO301, including ulcerative colitis.
About IL-18
IL-18 is a known inflammatory pathway which plays a key role in various immune processes and acts as a key upstream therapeutic target for AD and other chronic inflammatory diseases. As an alarmin cytokine, IL-18 acts as a general amplifier of inflammation, capable of driving multiple inflammatory responses, including Th1, Th2, Th17/22 and innate pathways. This upstream position is crucial for treating conditions with heterogeneous inflammation, where targeting a single pathway may not be optimal. An upregulated IL-18 pathway cyclically activates inflammatory mediators in an aberrant manner, resulting in tissue damage and other inflammatory and sensory disease pathology. IL-18 directly disrupts essential barrier functions, positioning it as a pivotal pathogenic factor. Targeting IL-18 offers a novel broader therapeutic utility, simultaneously reducing inflammation and restoring tissue integrity for patients with complex inflammatory conditions.
About EVO301
EVO301 is a long-acting injectable SAFA-IL-18BP fusion protein designed to neutralize aberrantly upregulated IL-18 activity. EVO301 facilitates more efficient tissue distribution and improved binding affinity and specificity relative to existing attempts to antagonize or inhibit the IL-18 pathway, including traditional mAbs. In addition, EVO301 incorporates several distinguishing design features, including selective and high binding affinity of native human IL-18BP and binding to serum albumin, smaller molecular weight, extended half-life for the neutralization of IL-18, and lower potential for immunogenicity. Evommune believes the distinct mechanism and modality of EVO301 complement those of EVO756, providing multiple, potentially synergistic avenues to bring innovative therapeutics to the large, underserved and rapidly expanding population of patients suffering from chronic inflammatory diseases.
About Evommune, Inc.
Evommune, Inc., is a clinical-stage biotechnology company developing innovative therapies that target key drivers of chronic inflammatory diseases. The Company’s mission is to improve patients’ daily lives and prevent the long-term effects of uncontrolled inflammation that are a consequence of the limitations of existing therapies. To achieve this, Evommune is advancing a portfolio of differentiated product candidates that target key drivers of chronic inflammation.