-
The positive opinion is based on data from the Phase 3 BRAVE-AA-PEDS study, in which 42% of patients with severe alopecia areata (AA) reached 80% or more scalp hair coverage at 36 weeks
-
The study is the first and largest of its kind specifically designed to evaluate children and adolescents with severe AA, a disease that has devastating social and emotional impact
-
Lilly has also submitted Olumiant in the U.S. for approval to treat severe AA in adolescents, with a decision expected in the second half of 2026
INDIANAPOLIS, Feb. 27, 2026 /PRNewswire/ -- Eli Lilly and Company (NYSE: LLY) and Incyte (NASDAQ: INCY) announced today that the European Medicines Agency's (EMA) Committee for Medicinal Products for Human Use (CHMP) has issued a positive opinion for Olumiant (baricitinib) for the treatment of adolescents (ages 12 to <18) with severe alopecia areata (AA). AA is a chronic immune disease that can have an especially devastating social and emotional impact on young patients and their families, as early onset AA can be more severe and lead to extensive and unpredictable hair loss.1
"The positive CHMP opinion supports the potential expansion of Olumiant as a new treatment option for adolescents living with the profound physical and emotional realities of severe alopecia areata," said Anabela Cardoso, senior vice president, Lilly Immunology Medical Affairs. "The depth and rigor of data from BRAVE-AA-PEDS – the first and largest trial designed specifically for these young patients – reflects Lilly's longstanding commitment to advancing care for people with chronic skin diseases who have had limited options for far too long."
Olumiant is a once-daily, oral JAK inhibitor discovered by Incyte and licensed to Lilly. In 2022, the European Commission (EC) and U.S. Food and Drug Administration (FDA) approved Olumiant for adult patients with severe AA, making it the first JAK inhibitor approved in these geographies for severe disease. This positive opinion marks the next step toward European regulatory approval of Olumiant for adolescents ages 12 to under 18 with severe AA, and it is now referred to the European Commission for final action. The European Commission's decision is expected in the next one to two months.
The positive CHMP opinion is supported by 36-week data from the Phase 3 BRAVE-AA-PEDS study evaluating the safety and efficacy of once-daily, oral Olumiant compared with placebo in the cohort of patients ages 12 to under 18. Treatment with Olumiant helped many adolescents achieve near-complete scalp hair regrowth. Additionally, many patients achieved successful eyebrow and eyelash regrowth.2
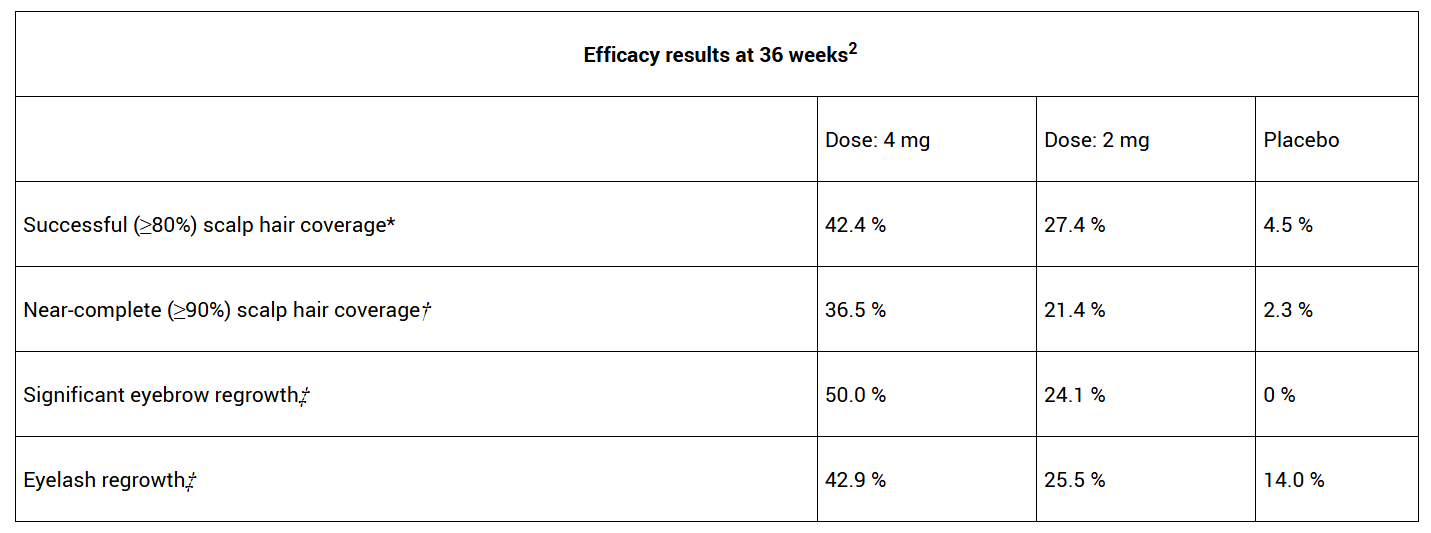
* Severity of Alopecia Tool (SALT) score ≤20
† SALT score ≤10
‡ Clinician-reported outcome (ClinRO) score of 0 or 1 with a ≥2-point improvement from baseline
The safety profile of Olumiant in adolescents with AA was consistent with the safety profile seen in clinical trials for children with juvenile idiopathic arthritis and moderate-to-severe atopic dermatitis.2
In addition, 52-week efficacy and safety data demonstrating successful hair regrowth on the scalp, eyebrows and eyelashes in adolescent patients were also recently presented at the Fall Clinical Dermatology (FCD) Conference in October 2025.
Olumiant is the most-researched JAK inhibitor in AA. In total for all indications, more than 14,600 patients have received Olumiant in completed and ongoing clinical trials. Of these, over 1,200 have been children and adolescents.
"Adolescents with severe alopecia areata represent a particularly vulnerable population, as the disease is difficult to manage and occurs at a time when appearance can have a significant impact on social identity and emotional well-being," said Thierry Passeron, M.D., PhD, professor and chair, Department of Dermatology, Université Côte d'Azur. "In clinical practice, families are frequently left with limited options that fall short. If approved in the European Union, Olumiant will represent an important evidence-based treatment option for these young patients — and with it, the hope that more adolescents living with severe alopecia areata can be helped."
Lilly has submitted Olumiant in the U.S. for approval to treat severe AA in adolescents, with a decision expected in the second half of 2026.
About BRAVE-AA-PEDS
BRAVE-AA-PEDS (NCT05723198) is an ongoing, placebo-controlled, Phase 3 clinical trial involving children and adolescents ages 6 to under 18 years with severe AA, as measured by a SALT score of ≥50 (i.e., who had ≥ 50% scalp hair loss) and a current episode of severe AA lasting at least six months but no more than eight years.
The first two cohorts of patients enrolled in BRAVE-AA-PEDS included adolescents (ages 12 to under 18 years, weighing ≥ 30 kg). The first cohort included 257 adolescent participants who were randomized in a 1:1:1 ratio to receive once-daily placebo, Olumiant 4 mg or Olumiant 2 mg. The primary endpoint of this study was a SALT score ≤20 (i.e., 80% or more scalp hair coverage) at Week 36. The second cohort of 166 adolescents were randomized 1:1 to Olumiant 4 mg or Olumiant 2 mg to further accumulate safety data.
The third cohort consists of children ages 6 to under 12 years and will be randomized in a 1:1:1 ratio to receive once-daily placebo, Olumiant high dose or Olumiant low dose.3
About Olumiant
Olumiant, a once-daily, oral JAK inhibitor, was discovered by Incyte and licensed to Lilly. Baricitinib is approved in the U.S. and more than 75 countries as a treatment for adults with moderately to severely active rheumatoid arthritis, in more than 40 countries outside the U.S. for the treatment of patients down to the age of two with moderate-to-severe atopic dermatitis who are candidates for systemic therapy and in the U.S., Europe and Japan for adult patients with severe AA. Marketing authorization for the treatment of hospitalized patients with COVID-19 has been granted for baricitinib in multiple countries.
The U.S. FDA-approved labeling for Olumiant includes a Boxed Warning for Serious Infections, Mortality, Malignancy, Major Adverse Cardiovascular Events, and Thrombosis. See the full Prescribing Information here.4
In December 2009, Lilly and Incyte announced an exclusive worldwide license and collaboration agreement for the development and commercialization of Olumiant and certain follow-on compounds for patients with inflammatory and autoimmune diseases.
About Lilly
Lilly is a medicine company turning science into healing to make life better for people around the world. We've been pioneering life-changing discoveries for nearly 150 years, and today our medicines help tens of millions of people across the globe. Harnessing the power of biotechnology, chemistry and genetic medicine, our scientists are urgently advancing new discoveries to solve some of the world's most significant health challenges: redefining diabetes care; treating obesity and curtailing its most devastating long-term effects; advancing the fight against Alzheimer's disease; providing solutions to some of the most debilitating immune system disorders; and transforming the most difficult-to-treat cancers into manageable diseases. With each step toward a healthier world, we're motivated by one thing: making life better for millions more people. That includes delivering innovative clinical trials that reflect the diversity of our world and working to ensure our medicines are accessible and affordable.
About Incyte
Incyte is a Wilmington, Delaware-based, global biopharmaceutical company focused on finding solutions for serious unmet medical needs through the discovery, development and commercialization of proprietary therapeutics.
References
-
Toussi, A., Barton, V. R., Le, S. T., Agbai, O. N., & Kiuru, M. (2021). Psychosocial and psychiatric comorbidities and health-related quality of life in alopecia areata: A systematic review. Journal of the American Academy of Dermatology, 85(1), 162–175. https://doi.org/10.1016/j.jaad.2020.06.047
-
Passeron T, et al. Baricitinib provides significant hair regrowth in adolescents with severe alopecia areata: 36-week efficacy and safety results from a Phase 3 randomized, controlled trial. American Academy of Dermatology Annual Meeting. March 7-11, 2025
-
Craiglow B, et al. Baricitinib provides significant hair regrowth in adolescents with severe alopecia areata: 52-week efficacy and safety results from a Phase 3 randomized, controlled trial. 2025 Fall Clinical Dermatology Conference. October 24, 2025
-
Olumiant. Prescribing Information. Lilly USA, LLC.