-
BioNTech on track for a catalyst-rich year with six late-stage data readouts expected across immunomodulators, antibody-drug conjugates and mRNA cancer immunotherapies
-
Increased focus on PD-L11/VEGF-A bispecific immunomodulator pumitamig with eight global Phase 3 clinical trials planned to be ongoing for year-end in collaboration with Bristol Myers Squibb
-
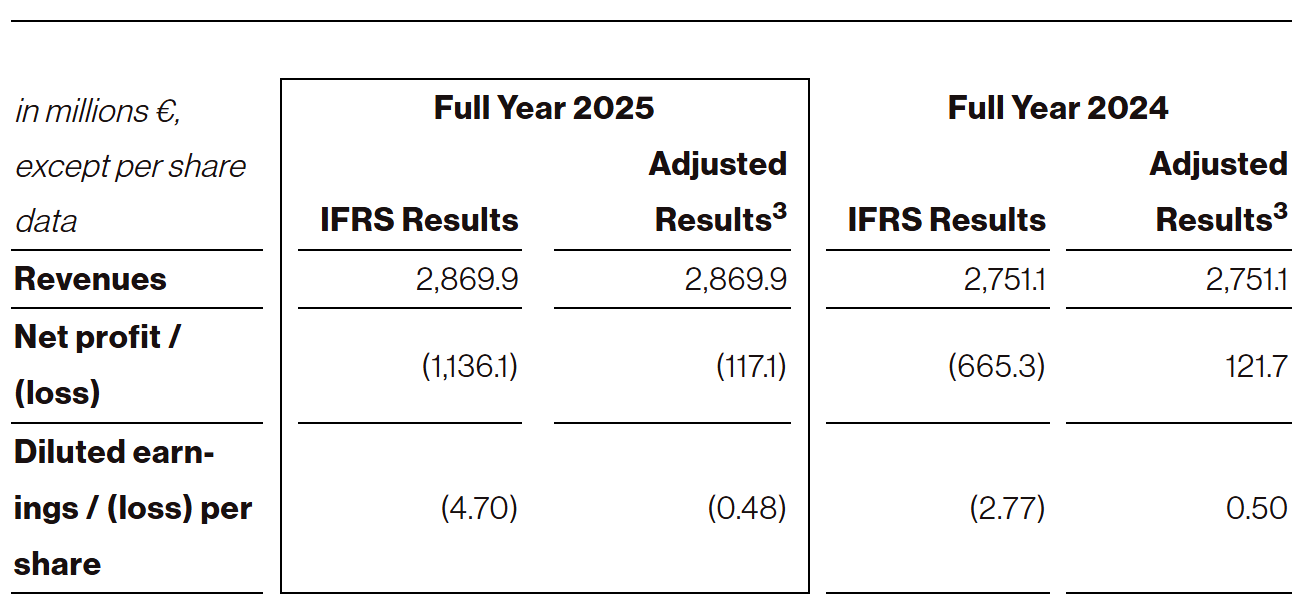
Full year 2025 revenues of €2.9 billion2, net loss of €1.1 billion (adjusted3 net loss of €0.1 billion) and diluted loss per share of €4.70 ($5.314) (adjusted3 diluted loss per share of €0.48 ($0.554))
-
Strong financial position continues to de-risk execution with cash, cash equivalents and security investments of €17.2 billion5
-
Expect 2026 total revenues of €2.0 billion to €2.3 billion, adjusted3 R&D expenses between €2.2-2.5 billion and adjusted3 SG&A expenses7 between €700-800 million
-
BioNTech co-founders Ugur Sahin and Özlem Türeci to establish an independent company with a focus on next-generation mRNA innovations; management transition by end of 2026
-
MAINZ, Germany, March 10, 2026 (GLOBE NEWSWIRE) -- BioNTech SE (Nasdaq: BNTX, “BioNTech” or “the Company”) today reported financial results for the three months and full year ended December 31, 2025 and provided an update on its corporate progress.
“2025 was a year of strong execution and pipeline momentum, marked by substantial progress in delivering on our strategy. We advanced our oncology pipeline by moving multiple programs into late-stage development and initiated trials assessing novel-novel combination approaches with the aim of delivering differentiated therapeutic profiles,” said Prof. Ugur Sahin, M.D., Chief Executive Officer and Co-Founder of BioNTech. “With our unique pipeline and strong financial position, we remain committed to leveraging our pioneering position in the immuno-oncology space with next-generation agents designed to elevate outcomes for patients with cancer. 2026 is poised to be a pivotal year with multiple readouts expected across our portfolio, representing a significant step toward our objective of becoming a multi-product company by 2030.”
Financial Review for Fourth Quarter and Full Year 20253
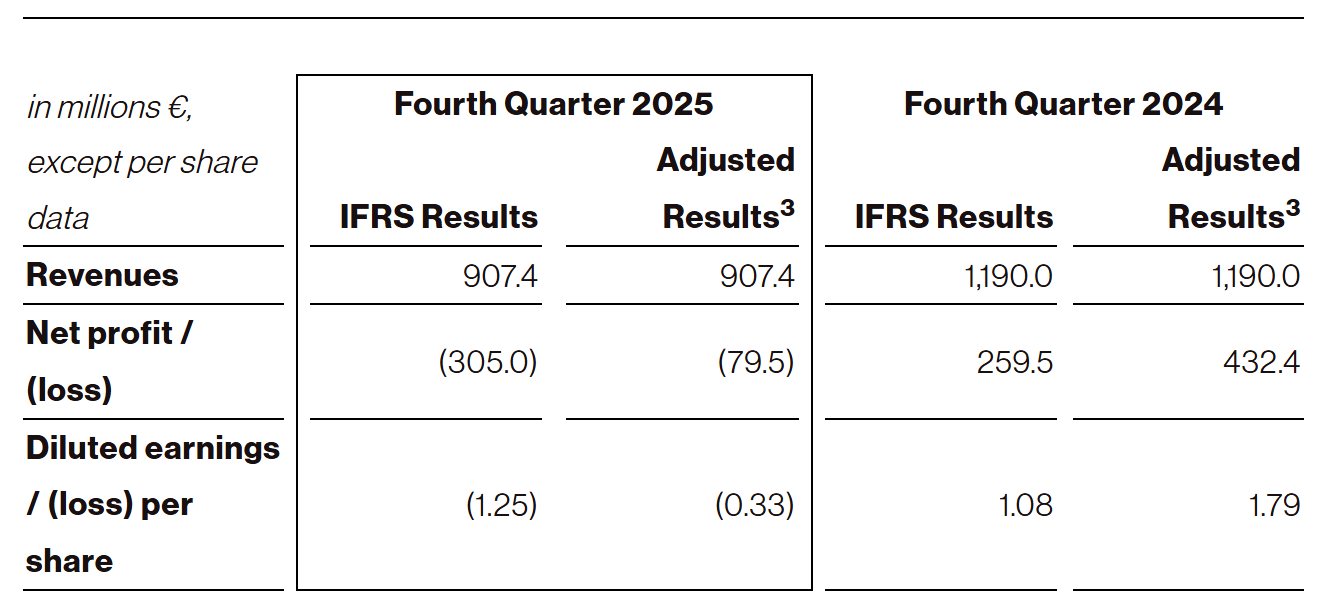
Revenues for the three months ended December 31, 2025 were €907.4 million, compared to €1,190.0 million for the comparative prior year period. For the year ended December 31, 2025, revenues were €2,869.9 million, compared to €2,751.1 million for the comparative prior year period. The quarterly year-on-year decrease was primarily driven by lower sales of the Company’s COVID-19 vaccines due to reduced market demand. The full year revenue increase was primarily driven by revenues related to BioNTech’s collaboration with Bristol Myers Squibb Company (“BMS”) that were recognized in the third quarter of 2025.
Research and development (“R&D”) expenses were €505.4 million for the three months ended December 31, 2025, compared to €611.8 million for the comparative prior year period. For the year ended December 31, 2025, R&D expenses were €2,104.9 million, compared to €2,254.2 million for the comparative prior year period. Both quarterly and full year year-on-year decreases were mainly driven by cost savings resulting from active portfolio management and positive effects resulting from our pumitamig cost sharing with BMS, partly offset by the acceleration of late-stage trials for immuno-oncology (“IO”) and antibody-drug conjugate (“ADC”) development programs.
Adjusted R&D expenses were €505.4 million for the three months ended December 31, 2025, compared to €530.3 million for the comparative prior year period. For the year ended December 31, 2025, adjusted R&D expenses were €2,019.5 million, compared to €2,172.7 million for the comparative prior year period. For 2025 and 2024, the Company’s adjusted R&D expenses exclude impairments.
Sales, general and administrative (“SG&A”) expenses7 were €217.9 million for the three months ended December 31, 2025, compared to €132.1 million for the comparative prior year period. For the year ended December 31, 2025, SG&A expenses were €624.4 million, compared to €599.0 million for the comparative prior year period. Both quarterly and full year year-on-year increases were mainly driven by our ongoing commercial build-up, partly offset by lower costs for external services.
Other operating result was negative €173.6 million during the three months ended December 31, 2025, compared to negative €54.0 million for the comparative prior year period. For the year ended December 31, 2025, other operating result was negative €903.7 million compared to negative €670.9 million for the prior year period. Both quarterly and full year year-on-year decreases were primarily driven by expenses from settlements of contractual disputes, expenses in connection with our pipeline prioritization and foreign exchange differences.
Adjusted other operating result was €21.4 million during the three months ended December 31, 2025, compared to negative €1.6 million for the comparative prior year period. For the year ended December 31, 2025, other operating result was negative €0.6 million compared to negative €13.5 million for the prior year period. For fiscal years 2025 and 2024, our quarterly and full year adjusted other operating results exclude expenses in connection with the settlements of legal proceedings. In addition, our quarterly and full year adjusted other operating results during fiscal year 2025 exclude employee-related costs in connection with our pipeline prioritization and a bargain purchase.
Net loss was €305.0 million for the three months ended December 31, 2025, compared to a net income of €259.5 million for the comparative prior year period. For the year ended December 31, 2025, net loss was €1,136.1 million, compared to a net loss of €665.3 million for the comparative prior year period.
Adjusted net loss was €79.5 million for the three months ended December 31, 2025, compared to an adjusted net profit of €432.4 million for the comparative prior year period. For the year ended December 31, 2025, adjusted net loss was €117.1 million, compared to an adjusted net profit of €121.7 million for the comparative prior year period.
Diluted loss per share was €1.25 for the three months ended December 31, 2025, compared to diluted earnings per share of €1.08 for the comparative prior year period. For the year ended December 31, 2025, diluted loss per share was €4.70, compared to diluted loss per share of €2.77 for the comparative prior year period.
Adjusted diluted loss per share was €0.33 for the three months ended December 31, 2025, compared to adjusted diluted earnings per share of €1.79 for the comparative prior year period. For the year ended December 31, 2025, adjusted diluted loss per share was €0.48, compared to adjusted diluted earnings per share of €0.50 for the comparative prior year period.
Cash, cash equivalents and security investments as of December 31, 2025 were €17,235.6 million, comprising €7,675.4 million in cash and cash equivalents, €7,158.5 million in current security investments disclosed as financial assets and €2,401.7 million in non-current security investments disclosed as financial assets.
Shares outstanding as of December 31, 2025 were 251,325,340, excluding 7,702,147 shares held in treasury.
“Our strong financial position fuels and de-risks our R&D activities as we prepare for multiple product launches in the coming years. Our financial discipline, active portfolio management and targeted investments will continue to drive innovation and create long-term value for BioNTech’s stakeholders,” said Ramón Zapata, Chief Financial Officer at BioNTech.
2026 Financial Year Guidance6:

In 2026, BioNTech anticipates lower COVID-19 vaccine revenues compared to 2025, driven by declines in both the European and United States markets. The United States continues to be a competitive and dynamic market, where as a result, lower revenues are expected. In Europe, we expect lower revenues as we defend our market share and begin managing the transition of multi-year contracts. In Germany, specifically, BioNTech recognizes direct sales of its COVID-19 vaccines as revenue. Hence, the anticipated declines in the Company’s sales of COVID-19 vaccines in the country will have a direct impact on its topline, whereas revenues outside of Germany only affect the Company’s topline as part of the 50% gross profit split with our partner Pfizer Inc. (“Pfizer”). Per the outlined partnership terms, revenues from the collaboration with BMS in 2026 are expected to be broadly in line with 2025. Revenues from the pandemic preparedness contract with the German government and service businesses are expected to remain stable.
Planned 2026 Financial Year Adjusted Expenses6:

BioNTech will continue to focus investments on R&D and scaling the business for late-stage development and commercial readiness in oncology, while remaining cost-disciplined. Strategic capital allocation will continue to foster innovation and be a key driver of the Company’s trajectory. As part of BioNTech’s strategy, the Company may continue to evaluate appropriate corporate development opportunities with the aim of driving sustainable long-term growth and creating future value.
The full audited consolidated financial statements as of and for the year ended December 31, 2025, can be found in BioNTech’s Annual Report on Form 20-F filed today with the U.S. Securities and Exchange Commission (“SEC”) and available at www.sec.gov.
Endnotes
1 An overview of abbreviations is compiled in a directory at the end of this press release.
2 All numbers in this press release have been rounded.
3 In addition to BioNTech’s results determined in accordance with International Financial Reporting Standards (“IFRS”), or IFRS Accounting Standards, or IFRS results, BioNTech reports certain adjusted, non-IFRS measures used internally as a supplemental measure of our business performance (each referred to with the prefix “Adjusted” or, as a whole, “Adjusted Results”). The calculation of these measures and the adjusted results as a whole is based on the concepts of the applicable IFRS Accounting Standards, but includes certain adjustments. Reconciliation of the adjusted results to BioNTech’s measures based on IFRS Accounting Standards and more information can be found at the end of this press release and in BioNTech’s Report on Form 20-F for the year ended December 31, 2025 filed on March 10, 2026, which is available at www.sec.gov. While non-IFRS measures may offer additional insights, BioNTech’s non-IFRS measures are not, and should not be viewed as, a substitute for their most directly comparable IFRS Accounting Standards measures, and should always be considered alongside our financial statements prepared in accordance with IFRS Accounting Standards.
4 Calculated applying the average foreign exchange rate for the year ended December 31, 2025, as published by the German Central Bank (Deutsche Bundesbank).
5 As of December 31, 2025.
6 Excludes risks that are not yet known and/or quantifiable and related activities. It includes effects identified from licensing arrangements, collaborations and Merger & Acquisitions (“M&A”) transactions to the extent disclosed. The guidance is based on non-IFRS measures and excludes certain effects compared to measures based on IFRS Accounting Standards. More information can be found in BioNTech’s Report on Form 20-F for the year ended December 31, 2025 filed on March 10, 2026, which is available at www.sec.gov.
7 Sales, general and administrative expenses (“SG&A”) include sales and marketing expenses as well as general and administrative expenses. Adjusted SG&A expenses include adjusted sales and marketing expenses as well as adjusted general and administrative expenses.
About BioNTech
Biopharmaceutical New Technologies (BioNTech) is a global next generation immunotherapy company pioneering novel investigative therapies for cancer and other serious diseases. BioNTech exploits a wide array of computational discovery and therapeutic modalities with the intent of rapid development of novel biopharmaceuticals. Its diversified portfolio of oncology product candidates aiming to address the full continuum of cancer includes mRNA cancer immunotherapies, next-generation immunomodulators and targeted therapies such as antibody-drug conjugates (ADCs) and innovative chimeric antigen receptor (CAR) T cell therapies. Based on its deep expertise in mRNA development and in-house manufacturing capabilities, BioNTech and its collaborators are researching and developing multiple mRNA vaccine candidates for a range of infectious diseases alongside its diverse oncology pipeline. BioNTech has established a broad set of relationships with multiple global and specialized pharmaceutical collaborators, including Bristol Myers Squibb, Duality Biologics, Genentech, a member of the Roche Group, Genmab, MediLink, OncoC4, Pfizer and Regeneron.