-
Late-breaking presentation of results from the Phase 3 CORALreef AddOn trial evaluating enlicitide decanoate, Merck’s investigational oral PCSK9 inhibitor, which may help address the cardiovascular (CV) epidemic
-
Data from the Phase 2 CADENCE trial of WINREVAIRTM (sotatercept-csrk) in patients with the syndrome of combined post- and precapillary pulmonary hypertension and heart failure with preserved ejection fraction (CpcPH-HFpEF) to be featured as late-breaking presentation
March 16, 2026--RAHWAY, N.J.--(BUSINESS WIRE)-- Merck (NYSE: MRK), known as MSD outside of the United States and Canada, today announced new clinical data from the company’s cardio-pulmonary pipeline will be presented at the American College of Cardiology’s Annual Scientific Session and Expo (ACC.26) in New Orleans, La., from March 28-30. The results shared will highlight Merck’s unwavering commitment to advancing innovative research in hypercholesterolemia and in the syndrome of combined post- and precapillary pulmonary hypertension and heart failure with preserved ejection fraction (CpcPH-HFpEF) to help address the significant burden of these diseases.
Key data being shared include two late-breaking presentations:
-
Positive results from the Phase 3 CORALreef AddOn trial evaluating the efficacy and safety of enlicitide, an investigational, once-daily oral proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitor versus ezetimibe, versus bempedoic acid and versus ezetimibe and bempedoic acid in adults with hypercholesterolemia who are treated with a statin;
-
Positive results from the Phase 2 CADENCE trial evaluating WINREVAIR™ (sotatercept-csrk) in adults with CpcPH-HFpEF.
“The data planned for presentation at ACC will underscore the strength and momentum of Merck’s cardio-pulmonary pipeline as we work to advance research that helps address the CV epidemic impacting millions with atherosclerosis, as well as CpcPH-HFpEF, a distinct syndrome with limited treatment options,” said Dr. Joerg Koglin, senior vice president, head of general and specialty medicine, global clinical development, Merck Research Laboratories. “Building on our history of bringing forward innovative therapies in heart and lung disease, we are proud to share research at ACC.26 that improves our understanding of these serious diseases and has the potential to help patients around the world.”
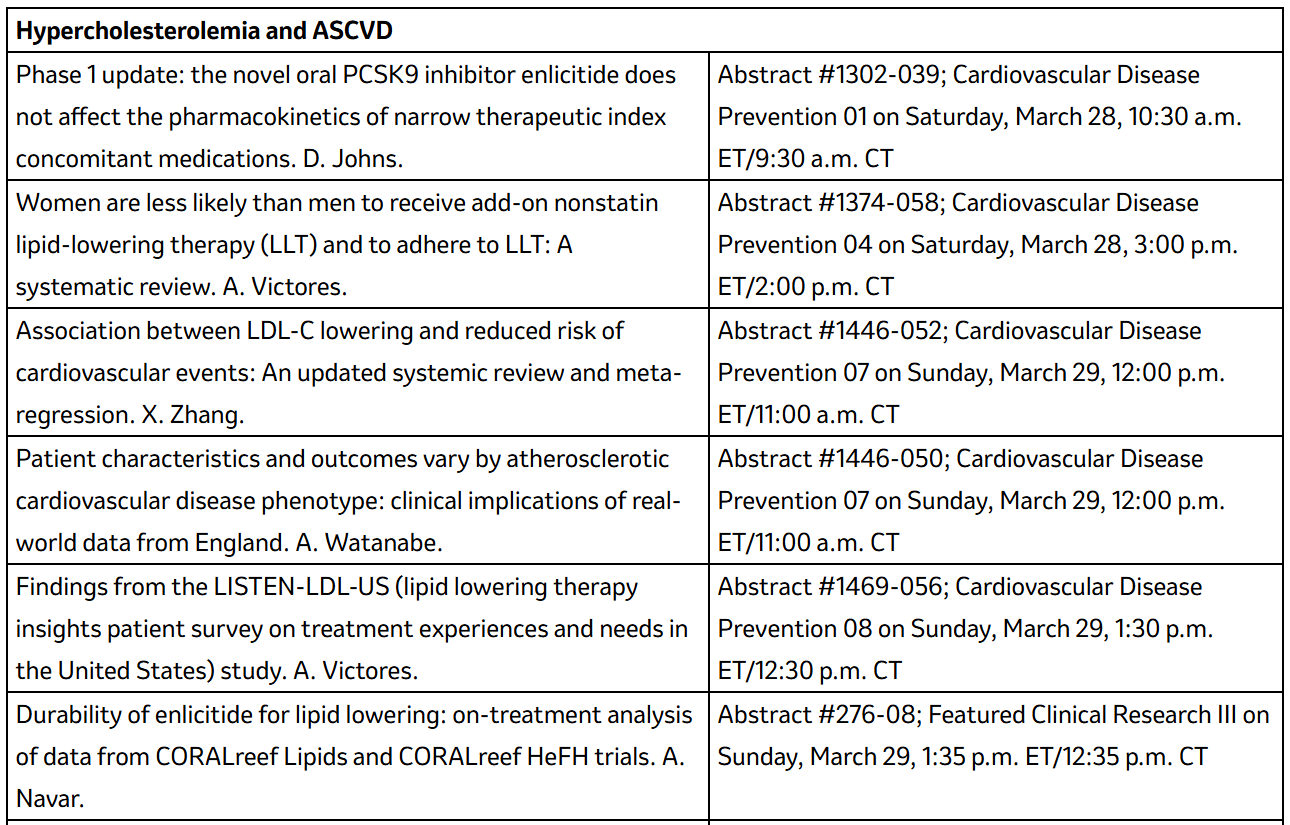
Details on Merck abstracts at ACC.26:

About enlicitide and PCSK9
Enlicitide has the potential to be the first approved oral PCSK9 inhibitor. It is designed to lower LDL-C via the same biological mechanism as currently approved monoclonal antibody, injectable PCSK9 inhibitors but in a daily pill form. Enlicitide is an investigational novel macrocyclic peptide that binds to PCSK9 and inhibits the interaction of PCSK9 with LDL receptors.
PCSK9 plays a key role in cholesterol homeostasis by regulating levels of the LDL receptor, which is responsible for the uptake of cholesterol into cells. Inhibition of PCSK9 is designed to prevent the interaction of PCSK9 with LDL receptors. This results in greater numbers of LDL receptors available on the cell surface to remove LDL cholesterol from the blood.
About CORALreef Clinical Trial Program
The efficacy and safety profile of enlicitide is being evaluated through the comprehensive CORALreef Clinical Trial program evaluating over 19,000 participants who have hypercholesterolemia. As previously announced, enlicitide demonstrated statistically significant and clinically meaningful reductions in LDL-C in three pivotal Phase 3 studies: CORALreef Lipids (NCT05952856), CORALreef HeFH (NCT05952869) and CORALreef AddOn (NCT06450366). Enlicitide is continuing to be evaluated in the large cardiovascular outcomes trial, CORALreef Outcomes (NCT06008756), which has completed enrollment with over 14,500 participants. Additional CORALreef clinical trials include CORALreef Extension (NCT06492291), CORALreef Pediatric (NCT07058077) and CORALreef Combination (NCT07216482).
About hypercholesterolemia
Hypercholesterolemia, a type of hyperlipidemia, is a disorder in which there are elevated LDL-C levels in the blood. It affects approximately 86 million adults in the U.S. and is a major risk factor for atherosclerotic cardiovascular disease (ASCVD). Nearly 70% of people with ASCVD who are treated with lipid lowering therapies do not reach target LDL cholesterol levels. High LDL-C, if left untreated, can lead to ASCVD events such as heart attacks and strokes.
About the CV epidemic and atherosclerotic cardiovascular disease
The silent CV epidemic is the leading cause of deaths globally, contributing to the majority of heart attacks and strokes, and deaths related to CV continue to rise. ASCVD accounts for 85% of CV deaths. It is caused by the buildup of plaque within the arteries, leading to narrowed or blocked blood vessels that can result in serious CV events such as heart attacks and strokes as well as coronary artery disease, peripheral artery disease and cerebrovascular disease.
About WINREVAIR™ (sotatercept-csrk) for injection, for subcutaneous use, 45 mg, 60 mg
In the U.S., WINREVAIR is FDA-approved for the treatment of adults with pulmonary arterial hypertension (PAH, WHO Group 1 pulmonary hypertension) to improve exercise capacity and World Health Organization (WHO) functional class (FC), and reduce the risk of clinical worsening events, including hospitalization for PAH, lung transplantation and death. WINREVAIR is the first activin signaling inhibitor therapy approved to treat PAH. WINREVAIR improves the balance between pro-proliferative and anti-proliferative signaling to modulate vascular proliferation. In preclinical models, WINREVAIR induced cellular changes that were associated with thinner vessel walls, partial reversal of right ventricular remodeling and improved hemodynamics.
WINREVAIR is the subject of a licensing agreement with Bristol Myers Squibb.
About the syndrome of combined post- and precapillary pulmonary hypertension and heart failure with preserved ejection fraction (CpcPH-HFpEF)
Combined post- and precapillary pulmonary hypertension and heart failure with preserved ejection fraction (CpcPH-HFpEF) is a distinct, identifiable and well-characterized condition that develops in people with long-term or advanced heart failure. Different from Group 1 pulmonary arterial hypertension (PAH), CpcPH-HFpEF is caused by two interrelated components: pulmonary vascular disease and cardiac disease. CpcPH-HFpEF is thought to be uncommon and underdiagnosed, typically impacting people who are older and have other comorbid conditions. It is associated with a worse prognosis and higher mortality rate compared to HFpEF alone. There are no treatments specifically approved for CpcPH-HFpEF.
Merck’s focus on cardiometabolic and respiratory diseases
Merck has a long history of developing treatments for cardiometabolic and respiratory diseases. Building on a legacy that began nearly 70 years ago with the introduction of our first cardiovascular therapy, we are committed to advancing research for patients impacted by cardiometabolic and respiratory diseases. Our focus spans a range of diseases, including atherosclerotic cardiovascular disease, heart failure, pulmonary hypertension and chronic obstructive pulmonary disease (COPD).
Advancements in the treatment of cardiometabolic and respiratory diseases can make a critical difference for patients and health systems around the world. At Merck, we strive for scientific excellence and innovation in all stages of research, from discovery through approval and life cycle management. We partner with experts in the community to advance research that can help improve the lives of patients.
About Merck
At Merck, known as MSD outside of the United States and Canada, we are unified around our purpose: We use the power of leading-edge science to save and improve lives around the world. For more than 130 years, we have brought hope to humanity through the development of important medicines and vaccines. We aspire to be the premier research-intensive biopharmaceutical company in the world – and today, we are at the forefront of research to deliver innovative health solutions that advance the prevention and treatment of diseases in people and animals. We foster a diverse and inclusive global workforce and operate responsibly every day to enable a safe, sustainable and healthy future for all people and communities.