-
Fabhalta lowered likelihood of progression to kidney failure by 43% in APPLAUSE-IgAN study1
-
40.7% of patients on Fabhalta demonstrated sustained reduction of protein in urine over two years1
-
Fabhalta granted priority review by FDA for traditional approval
Basel, March 29, 2026 – Novartis today announced final two-year results from the Phase III APPLAUSE‑IgAN study of Fabhalta® (iptacopan) in IgA nephropathy (IgAN). Fabhalta demonstrated a statistically significant, clinically meaningful improvement in estimated glomerular filtration rate (eGFR) slope, a key marker of kidney function, compared with placebo1. Fabhalta consistently outperformed placebo across key kidney outcomes over two years, demonstrating a slowing of disease progression and the potential to preserve kidney function in IgAN1.
The results were published in the New England Journal of Medicine and simultaneously presented as late‑breaking data at the 2026 World Congress of Nephrology (WCN).
“Persistent kidney inflammation is a hallmark of IgAN, and a key driver of disease progression, leading to ongoing kidney damage and loss of function over time,” said Vlado Perkovic, MD, Professor of Medicine and Provost, University of New South Wales, and Steering Committee Co‑Chair of the APPLAUSE‑IgAN study. “These results are important because they show that Fabhalta can reduce the risk of disease progression, help preserve kidney health, and address outcomes associated with long-term disease burden.”
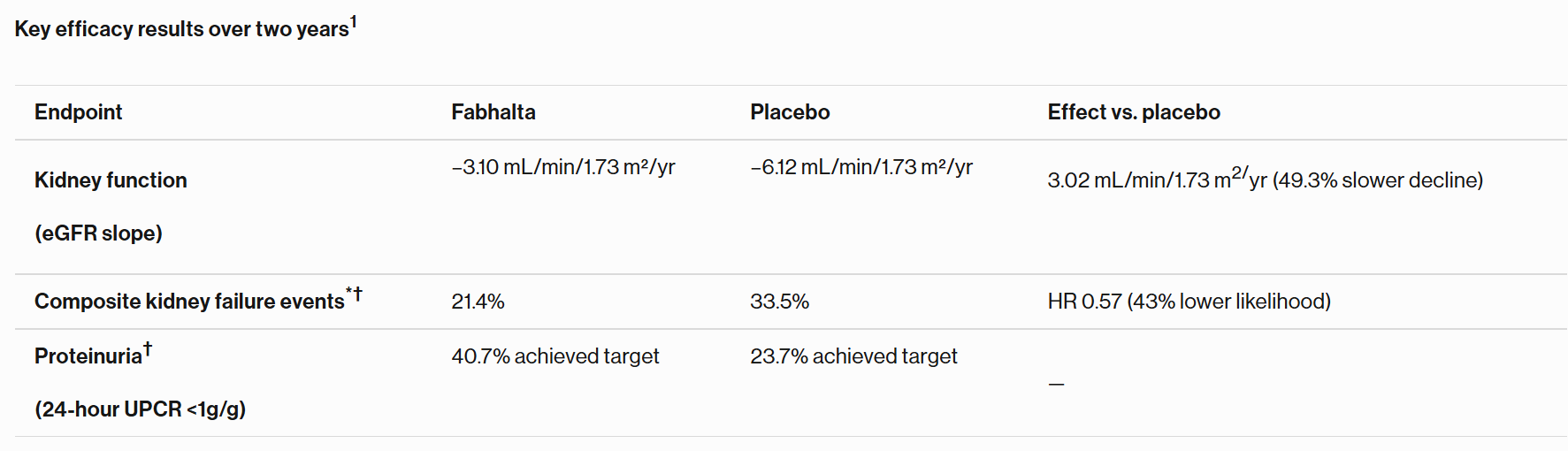
*Composite kidney failure endpoint: reaching either sustained ≥30% decline in eGFR relative to baseline, sustained eGFR <15 mL/min/1.73 m², initiation of maintenance dialysis, kidney transplant, or death from kidney failure
†As measured by percentage of patients
“The two-year results demonstrate that Fabhalta consistently and meaningfully slows kidney function decline in high-risk patients with IgAN,” said Ruchira Glaser, MD, MS, Global Head, Cardiovascular, Renal and Metabolic Development, Novartis. “This progress reflects years of focused research and supports our efforts to advance more targeted treatment options to help preserve kidney health in people living with IgAN.”
The safety profile of Fabhalta over two years was consistent with previous findings. Rates of adverse events and treatment discontinuation were low and similar between Fabhalta and placebo1,2.
Fabhalta received accelerated approval in the U.S. and China for proteinuria reduction in adults with IgAN based on data from a prespecified interim analysis of the APPLAUSE-IgAN study2,3. The two-year data were submitted to the U.S. Food and Drug Administration for traditional approval. Fabhalta was granted priority review based on the novel mode of action and the strength of the data. Alongside Fabhalta, Novartis continues to advance its multi-asset IgAN portfolio, which also includes Vanrafia® (atrasentan) and investigational compound zigakibart.
About IgAN
IgAN is a progressive autoimmune kidney disease, with approximately 25 people per million worldwide newly diagnosed each year4,5. IgAN is highly debilitating as it leads to inflammation in the small filters of the kidneys, excess protein in urine, and a gradual decline in eGFR6. Up to 50% of patients with persistent proteinuria progress to kidney failure within 10 to 20 years of diagnosis, often requiring dialysis or kidney transplantation as part of long-term disease management5-10.
Furthermore, people living with IgAN often face mental and social challenges6-9. Supportive care has not addressed the underlying causes of the disease and often fails to slow disease progression, reinforcing the need for more targeted therapies for IgAN7-12.
About APPLAUSE-IgAN
APPLAUSE-IgAN (NCT04578834) is a global, randomized, double-blind, placebo-controlled Phase III study evaluating Fabhalta in adults with biopsy-confirmed IgAN and persistent proteinuria despite optimized supportive care. Patients were randomized 1:1 to receive Fabhalta or placebo and were followed for up to 24 months11. The primary endpoint was the annualized total eGFR slope over 24 months. Key secondary endpoints included time to first composite kidney failure event and changes in proteinuria over 9 months1.
The most common adverse events with Fabhalta were mainly mild-to-moderate infections (such as COVID-19 and upper respiratory tract infection), headache, diarrhea, and hyperlipidemia, with overall adverse event rates comparable to placebo1.
About Fabhalta® (iptacopan)
Fabhalta (iptacopan) is an oral Factor B inhibitor designed to selectively target the alternative complement pathway, one of several key drivers of inflammation and kidney damage in IgAN4,12,13. By inhibiting Factor B, Fabhalta aims to reduce ongoing complement-mediated injury and slow disease progression. Fabhalta has received regulatory approvals in multiple complement-mediated diseases, including IgAN, and is being evaluated across a range of rare kidney conditions.
About Novartis
Novartis is an innovative medicines company. Every day, we work to reimagine medicine to improve and extend people’s lives so that patients, healthcare professionals and societies are empowered in the face of serious disease. Our medicines reach more than 300 million people worldwide.
References
-
Novartis. Data on file
-
Novartis Pharmaceuticals Corporation. Novartis receives FDA accelerated approval for Fabhalta® (iptacopan), the first and only complement inhibitor for the reduction of proteinuria in primary IgA nephropathy (IgAN) (2024). Available at: https://www.novartis.com/news/media-releases/novartis-receives-fda-accelerated-approval-fabhalta-iptacopan-first-and-only-complement-inhibitor-reduction-proteinuria-primary-iga-nephropathy-igan. Accessed March 2026
-
Novartis China. Novartis’ innovative medicine Fabhalta® (iptacopan) approved in China for a new indication. Novartis China website (in Mandarin). Available at: https://www.novartis.com.cn/news/nuohuazaiyingshenzangjibingzhiliaolingyulichengbeichuangxinyaowufeihedayansuanyipukepanjiaonangigashenbingshiyingzhengzaizhongguohuopi. Accessed March 2026
-
Rizk DV, Maillard N, Julian BA, et al. The emerging role of complement proteins as a target for therapy of IgA nephropathy. Front Immunol 2019;10:504.
-
Cheung C, Barratt J. The rapidly changing treatment landscape of IgA nephropathy. Semin Nephrol. 2025;44:151573.
-
Kwon CS, Daniele P, Forsythe A et al. A systematic literature review of the epidemiology, health-related quality of life impact, and economic burden of immunoglobulin a nephropathy. J Health Econ Outcomes Res. 2021;8:36–45.
-
Pitcher D, Braddon F, Hendry B et al. Long-term outcomes in IgAN. Clin J Am Soc Nephrol. 2023;18:727–8.
-
Mohd R, Mohammad Kazmin NE, Abdul Cader R, et al. Long-term outcome of immunoglobulin A (IgA) nephropathy: a single center experience. PLoS One.
-
National Kidney Foundation. The voice of the patient (2020). Available at: https://igan.org/wp-content/uploads/2021/01/VOP_IgAN_12-7-20__FNL.pdf. Accessed March 2026.
-
Kidney Disease: Improving Global Outcomes (KDIGO) Glomerular Diseases Work Group. KDIGO 2021 Clinical Practice Guideline for the Management of Glomerular Diseases. Kidney Int. 2021;100:S1–276.
-
Clinicaltrials.gov. NCT04578834. Study of Efficacy and Safety of LNP023 in Primary IgA Nephropathy Patients (APPLAUSE-IgAN). Available at: https://clinicaltrials.gov/study/NCT04578834. Accessed March 2026.
-
Perkovic V, Barratt J, Rovin B, et al. Alternative complement pathway inhibition with iptacopan in IgA nephropathy. N Engl J Med. 2025;392:531–543.
-
Chiu YL, Lin WC, Shu KH, et al. Alternative complement pathway is activated and associated with galactose-deficient IgA(1) antibody in IgA nephropathy patients. Front Immunol 2021;12:638309.