-
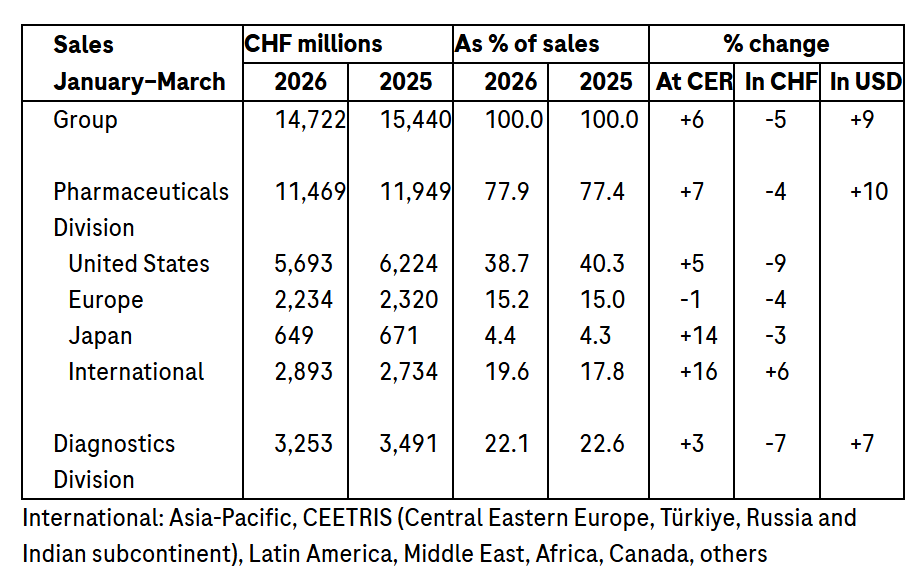
Group sales were +6% at constant exchange rates (CER)1, -5% when reported in CHF and +9% in USD2, in the first three months, driven by high demand for our innovative medicines and diagnostics.
-
Pharmaceuticals Division sales were +7% at CER, -4% when reported in CHF and +10% in USD, due to continued high growth in sales of medicines for the treatment of severe diseases; Xolair (chronic hives, food allergies), Phesgo (breast cancer), Hemlibra (haemophilia A), Vabysmo (severe eye diseases) and Ocrevus (multiple sclerosis) were the top growth drivers.
-
Diagnostics Division sales were +3% at CER, -7% when reported in CHF and +7% in USD, as demand for core lab and pathology solutions more than offset the impact of healthcare pricing reforms in China.
-
Highlights:
Positive data for: fenebrutinib for multiple sclerosis (phase III), Gazyva/Gazyvaro for an autoimmune condition affecting kidney function (phase III); and petrelintide for obesity (phase II)
CE mark for new Elecsys NfL blood test to detect neuroinflammation in multiple sclerosis, representing a breakthrough in disease management
Launch of the cobas MPX-E assay, a new 4-in-1 test to screen blood donors for HIV and hepatitis viruses
Launch of the AI factory to accelerate the development of new therapeutics and diagnostics solutions
Entry into a definitive agreement to acquire SAGA Diagnostics: SAGA’s proprietary cancer therapy response monitoring platform will strengthen Roche’s industry leading portfolio of oncology diagnostics and medicines
Inauguration of the new research home for the Institute of Human Biology to enable scientists to pioneer human model systems for research
Outlook for 2026 confirmed
Roche CEO Thomas Schinecker: “We delivered a strong start to the year, achieving 6% Group sales growth at constant exchange rates.
Our pipeline continued to advance in areas where patients face significant unmet need, including multiple sclerosis, obesity and a severe autoimmune disease that can lead to kidney failure. We also received EU approval for a new test to detect neuroinflammation in multiple sclerosis, marking a meaningful step forward in disease management.
Our diversified portfolio across both divisions, together with continued pipeline progress, positions us well for sustained future growth in a dynamic geopolitical environment. We confirm our full-year outlook.”
-
Outlook for 2026
April 23, 2026--Roche CEO Thomas Schinecker: “We delivered a strong start to the year, achieving 6% Group sales growth at constant exchange rates.
Our pipeline continued to advance in areas where patients face significant unmet need, including multiple sclerosis, obesity and a severe autoimmune disease that can lead to kidney failure. We also received EU approval for a new test to detect neuroinflammation in multiple sclerosis, marking a meaningful step forward in disease management.
Our diversified portfolio across both divisions, together with continued pipeline progress, positions us well for sustained future growth in a dynamic geopolitical environment. We confirm our full-year outlook.”
Outlook for 2026
Roche (SIX: RO, ROP; OTCQX: RHHBY) expects an increase in Group sales in the mid single digit range (CER) for 2026. Core earnings per share are targeted to develop in the high single digit range (CER). Roche expects to further increase its dividend in Swiss francs.
Group sales
In the first three months of 2026, Roche sales were +6% at CER, -5% when reported in CHF, to CHF 14.7 billion due to strong demand for pharmaceutical products and diagnostic solutions. The appreciation of the Swiss franc against most currencies, notably the US dollar, had a significant impact on the results reported in Swiss francs compared to constant exchange rates.
Sales in the Pharmaceuticals Division were +7% at CER, -4% when reported in CHF, to CHF 11.5 billion, with medicines for severe diseases continuing their strong growth.
The top five growth drivers – Xolair, Phesgo, Hemlibra, Vabysmo and Ocrevus – achieved total sales of CHF 5.3 billion, an increase of 14% at CER, or 2% in CHF, compared to the first three months of 2025.
Sales of products with expired patents (Avastin, Herceptin, MabThera/Rituxan, Lucentis and Actemra/RoActemra) decreased by a combined CHF 0.1 billion at CER, or CHF 0.2 billion in CHF.
In the United States, sales were +5% at CER, -9% when reported in CHF, due to continued growth of Xolair and continuing uptake of Hemlibra, Polivy (blood cancer), Ocrevus and Vabysmo. This growth more than compensated for the lower sales of Perjeta (breast cancer) and Kadcyla (breast cancer).
Sales in Europe were -1% at CER, or -4% when reported in CHF, due to lower sales of Polivy, the impact of biosimilar competition on Actemra/RoActemra and Perjeta (breast cancer) due to conversion to Phesgo. This decrease was partially offset by the demand for Ocrevus and Evrysdi (spinal muscular atrophy).
In Japan, sales were +14% at CER, -3% when reported in CHF, mainly due to product supply to third parties, as well as a strong uptake of Hemlibra, Vabysmo and Polivy. Sales growth was partially offset by the decline in sales of Tamiflu and the impact of biosimilar erosion on Avastin.
Sales in the International region were +16% at CER, or +6% when reported in CHF, led by Phesgo, Vabysmo, Polivy, Alecensa (lung cancer) and Ocrevus and partially offset by lower sales of influenza medicines Xofluza and Tamiflu. In China, sales were +14% at CER, or +5% when reported in CHF, as the uptake of Phesgo, Polivy and Vabysmo gained momentum following their inclusion in the government drug reimbursement list. The continued roll-out of Alecensa was the other growth driver. The increase was partially offset by lower sales of Xofluza and Perjeta.
The Diagnostics Division’s sales were +3% at CER, -7% when reported in CHF, to CHF 3.3 billion as growth in demand for core lab and pathology solutions more than offset the impact of healthcare pricing reforms in China.
Sales in the Europe, Middle East and Africa (EMEA) region were +3% at CER, -2% when reported in CHF, driven by higher sales of clinical chemistry and immunodiagnostic products. In North America, sales were +6% at CER, -7% when reported in CHF, with growth across core, pathology and molecular lab areas. Sales in Asia-Pacific were -5% at CER, or -15% when reported in CHF, due to the healthcare pricing reforms in China. In Latin America, sales growth was +10% at CER, or 0% when reported in CHF.
About Roche
Roche (SIX: RO, ROP; OTCQX: RHHBY) is a healthcare company uniquely placed to prevent, stop and cure diseases by uniting leading science and technology across diagnostics, medicines and digital solutions.
Roche was founded in Basel, Switzerland in 1896 and today is a leading provider of transformative medicines and diagnostics for millions of people in over 150 countries around the world. It is dedicated to tackling healthcare challenges that place the greatest strain on patients, families, communities and healthcare systems. Across its Diagnostics and Pharmaceutical divisions, Roche focuses on areas including oncology, neurology, cardiovascular and metabolic diseases, ophthalmology, infectious diseases and immunology with the aim of providing real and positive change for patients, the people they love and the professionals who care for them.
Genentech in the United States is a fully owned subsidiary in the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, a major innovator in the Japanese therapeutic antibody market.
References
[1] CER (Constant Exchange Rates). The percentage changes at constant exchange rates are calculated using simulations by reconsolidating both the 2026 and 2025 results at constant exchange rates (the average rates for the year ended 31 December 2025). For the definition of CER, see page 178 of the Roche Finance Report 2025.
[2] USD (US dollars). The percentage changes for selected sales figures at US dollars are calculated by translating both the 2026 and 2025 sales figures at the respective average US dollar exchange rate for the period in question. This supplementary information is provided to assist readers when assessing comparability with other companies.
[3] Products launched before 2015.