-
Submission is supported by data from the Phase 3 UP-AA clinical program in which upadacitinib achieved the primary endpoint of severity of alopecia tool (SALT) score ≤ 20 at week 24, with additional efficacy results observed at week 521-3
-
Upadacitinib is also the first JAK inhibitor to meet the ranked secondary endpoint of complete scalp hair regrowth (SALT = 0) at week 241-3
NORTH CHICAGO, Ill., April 28, 2026 /PRNewswire/ -- AbbVie (NYSE: ABBV) today announced that it has submitted an application for a new indication to the U.S. Food and Drug Administration (FDA) for upadacitinib (RINVOQ®; 15 mg and 30 mg, once daily) for the treatment of adult and adolescent patients with severe alopecia areata (AA), based on results from the Phase 3 UP-AA clinical program.1-3
"Alopecia areata is a chronic immune-mediated disease in which the physical and psychological burden patients can experience goes far beyond hair loss, impacting their day-to-day lives," said Kori Wallace, M.D., Ph.D., vice president, global head of immunology clinical development, AbbVie. "In the UP-AA clinical program, upadacitinib showed early and substantial scalp hair growth, including complete scalp hair coverage, which is a significant outcome for those living with this often-overlooked disease."
UP-AA Clinical Trial Summary1-3
UP-AA includes two replicate studies with Period A as a double-blind, placebo-controlled phase for 24 weeks, followed by Period B, a blinded extension through 52 weeks. Results from Period B are summarized descriptively.
The mean baseline SALT score across the trials was 84 and approximately 51% of patients (n=716 out of 1,399) had SALT score ≥ 95 at baseline, representing a population with near-total or total scalp hair loss. SALT ≤ 20 (primary endpoint) and SALT = 0 (key ranked secondary endpoint) were met at week 24 for both upadacitinib doses (15 mg and 30 mg) and response rates improved through week 52 as shown below.
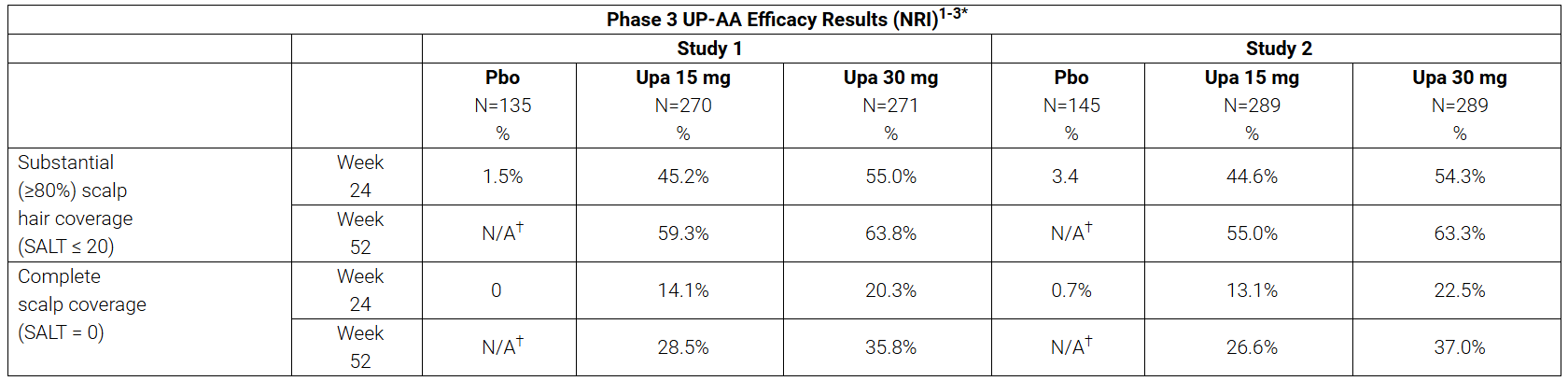
*The primary endpoint was SALT score ≤ 20 at week 24, and a key ranked secondary endpoint was SALT score = 0 at week 24 from the double-blind, placebo-controlled Period A. Data at week 52 are descriptive from the blinded extension Period B. SALT score ≤ 20 indicates ≥ 80% scalp hair coverage. SALT score = 0 indicates complete scalp hair coverage. †Pbo-controlled groups ended at week 24. NRI: Non-responder imputation. Pbo: Placebo.
The safety profile of upadacitinib 15 mg and 30 mg through week 52 was consistent with that observed at week 24 and shared in previously announced results.1-3
Use of upadacitinib in AA is not approved; its safety and efficacy are under regulatory review by the U.S. FDA and the European Medicines Agency.
About UP-AA Clinical Trial1-3
UP-AA M23-716 was conducted as a single protocol that includes two replicate pivotal studies (Study 1 and Study 2) with randomization, investigative sites, data collection, analysis and reporting independent for each study. These Phase 3 randomized, placebo-controlled, double-blind studies evaluate the efficacy and safety of upadacitinib in adult and adolescent patients with severe alopecia areata. In Study 1 and Study 2 Period A, participants were randomized to one of three groups to receive upadacitinib 15 mg, upadacitinib 30 mg or placebo for 24 weeks. In Study 1 and Study 2 Period B, participants originally randomized to upadacitinib dose groups in Period A continued their same treatment in Period B for 28 additional weeks. Participants originally randomized to placebo in Period A either remained on placebo in Period B or were re-randomized to either upadacitinib 15 or upadacitinib 30 mg, based on their SALT score at week 24. In total, Study 1 and Study 2 Periods A and B span 52 weeks. Results from Period B are summarized descriptively. Participants who complete Study 1 or Study 2 can join Study 3 and may be re-randomized to receive 1 of 2 doses of upadacitinib for up to 108 weeks, where data is summarized descriptively. The two trials randomized 1,399 participants with severe AA ages 12 to 63 across 248 sites worldwide. More information on this trial can be found at www.clinicaltrials.gov (NCT06012240).
About Alopecia Areata
Alopecia areata (AA) is an unpredictable autoimmune disease causing a range of hair loss patterns, from sudden, round bald patches on the scalp to complete loss of all body hair, including scalp, face, eyebrows and eyelashes.4,5 Despite its immune-mediated nature, AA is often considered a cosmetic problem, which can lead to stigma and have an impact on patients' lives.6,7
About RINVOQ® (upadacitinib)
Discovered and developed by AbbVie scientists, RINVOQ is a JAK inhibitor that is being studied in several immune-mediated inflammatory diseases. Based on enzymatic and cellular assays, RINVOQ demonstrated greater inhibitory potency for JAK-1 vs JAK-2, JAK-3, and TYK-2. The relevance of inhibition of specific JAK enzymes to therapeutic effectiveness and safety is not currently known.
Upadacitinib (RINVOQ) is being studied in Phase 3 clinical trials for alopecia areata, hidradenitis suppurativa, Takayasu arteritis, systemic lupus erythematosus, and vitiligo. The use of upadacitinib in alopecia areata is not approved; its safety and efficacy are under regulatory review by the U.S. FDA and the European Medicines Agency.
About AbbVie
AbbVie's mission is to discover and deliver innovative medicines and solutions that solve serious health issues today and address the medical challenges of tomorrow. We strive to have a remarkable impact on people's lives across several key therapeutic areas including immunology, neuroscience and oncology – and products and services in our Allergan Aesthetics portfolio.
References
-
AbbVie. Data on file ABVRRTI81580.
-
AbbVie. Data on file ABVRRTI81456.
-
AbbVie. Data on file ABVRRTI83115.
-
Alkhalifah A, Alsantali A, Wang E, McElwee KJ, Shapiro J. Alopecia areata update: Part I. Clinical picture, histopathology, and pathogenesis. J Am Acad Dermatol. 2010;62(2):177-188, quiz 189-190.
-
Pratt CH, King LE, Messenger AG, Christiano AM, Sundberg JP. Alopecia areata. Nat Rev Dis Primers. 2017;3(1):17011
-
Davey L, Clarke V, Jenkinson E. Living with alopecia areata: an online qualitative survey study. Br J Dermatol. 2019;180(6):1377-1389
-
Bain KA, McDonald E, Moffat F, et al. Alopecia areata is characterized by dysregulation in systemic type 17 and type 2 cytokines, which may contribute to disease-associated psychological morbidity. Br J Dermatol. 2020;182(1):130-137
-
RINVOQ [Package Insert]. North Chicago, IL: AbbVie Inc.; 2025