-
Five-year follow-up data from KEYNOTE-942 underscore the continued potential of intismeran autogene (mRNA-4157 or V940) in combination with KEYTRUDA® (pembrolizumab) for patients with resected high-risk melanoma
-
Results from the final analysis of KEYNOTE-522, evaluating KEYTRUDA in combination with chemotherapy, demonstrate continued survival benefit for patients with high-risk early-stage triple-negative breast cancer (TNBC)
-
New data for sacituzumab tirumotecan (sac-TMT), an investigational TROP2‑directed antibody‑drug conjugate, add to ongoing research of novel treatment approaches for patients with non‑small cell lung cancer
-
Data from ASCENT-04/KEYNOTE-D19, evaluating KEYTRUDA plus Trodelvy® (sacituzumab govitecan-hziy) for patients with metastatic TNBC, will be featured in the official ASCO 2026 Press Program
May 12, 2026--RAHWAY, N.J.--(BUSINESS WIRE)-- Merck (NYSE: MRK), known as MSD outside of the United States and Canada, today announced new research from more than 100 abstracts across over 25 types of cancer from the company’s comprehensive oncology portfolio and pipeline will be presented at the 2026 American Society of Clinical Oncology (ASCO) Annual Meeting (May 29-June 2). The data reinforce the long-term impact of KEYTRUDA® (pembrolizumab), Merck’s anti-PD-1 therapy, and Merck’s rapidly advancing pipeline across multiple tumor types and stages of disease, highlighting the company’s leadership in oncology and commitment to advancing innovative oncology research.
“During ASCO, we will present data that showcase the strong momentum in our oncology pipeline, including long-term data for intismeran autogene, our investigational individualized neoantigen therapy (INT),” said Dr. Marjorie Green, senior vice president and head of oncology global clinical development, Merck Research Laboratories. “We look forward to sharing new research for our oncology medicines and novel treatment approaches, such as our INT and antibody-drug conjugates, which may help address significant unmet medical needs for patients living with cancer.”
Key data from Merck’s portfolio and pipeline to be presented:
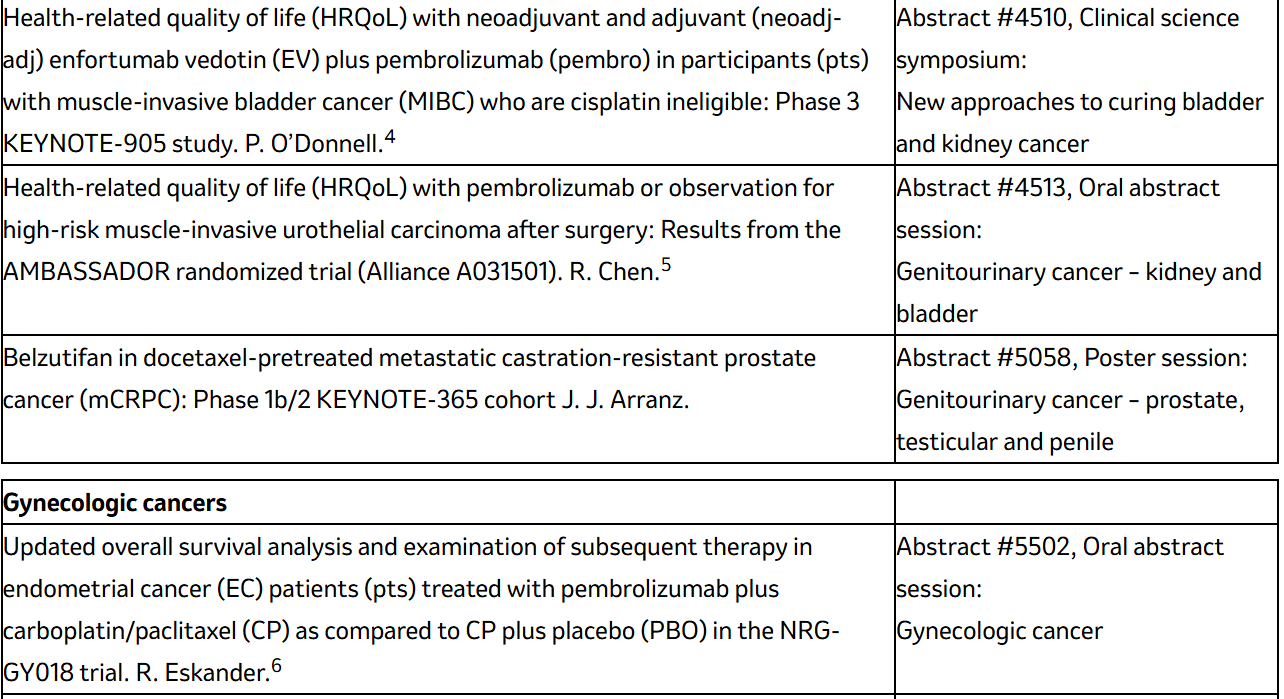
Five-year follow-up data from the Phase 2b KEYNOTE-942 trial evaluating intismeran autogene in combination with KEYTRUDA for patients with high-risk melanoma following complete resection (Abstract #9500, Oral abstract session: Melanoma/skin cancers). 1
Final analysis results from the Phase 3 KEYNOTE-522 trial evaluating KEYTRUDA in combination with chemotherapy as pre-operative treatment and then continuing as a single agent after surgery for the treatment of patients with high-risk early-stage triple-negative breast cancer (TNBC) (Abstract #507, Oral abstract session: Breast cancer – local/regional/adjuvant).
Data from the Phase 3 OptiTROP-Lung05 trial conducted in China, led by Kelun-Biotech, evaluating sac-TMT plus KEYTRUDA in advanced non-small cell lung cancer (Abstract #8506, Oral abstract session: Lung cancer – non-small cell metastatic). 2
Progression-free survival data from the Phase 3 ASCENT-04/KEYNOTE-D19 study evaluating KEYTRUDA plus Trodelvy (sacituzumab govitecan-hziy) in previously untreated PD-L1-positive metastatic TNBC (Abstract #LBA1000, Oral abstract session: Breast cancer – metastatic). 3
Merck investor event
Merck will hold an Oncology Investor Event to coincide with the 2026 ASCO Annual Meeting on Monday, June 1, 2026, 6 p.m. CT, during which senior management will provide an update on the company’s oncology strategy and program. The event will take place in Chicago, Ill., and will be accessible via webcast. Investors, analysts, members of the media and the general public are invited to listen to a webcast of the presentation via this weblink. All participants may join the call by dialing (800) 369-2154 (U.S. and Canada Toll-Free) or (517) 308-9422 and using the access code 8711041.
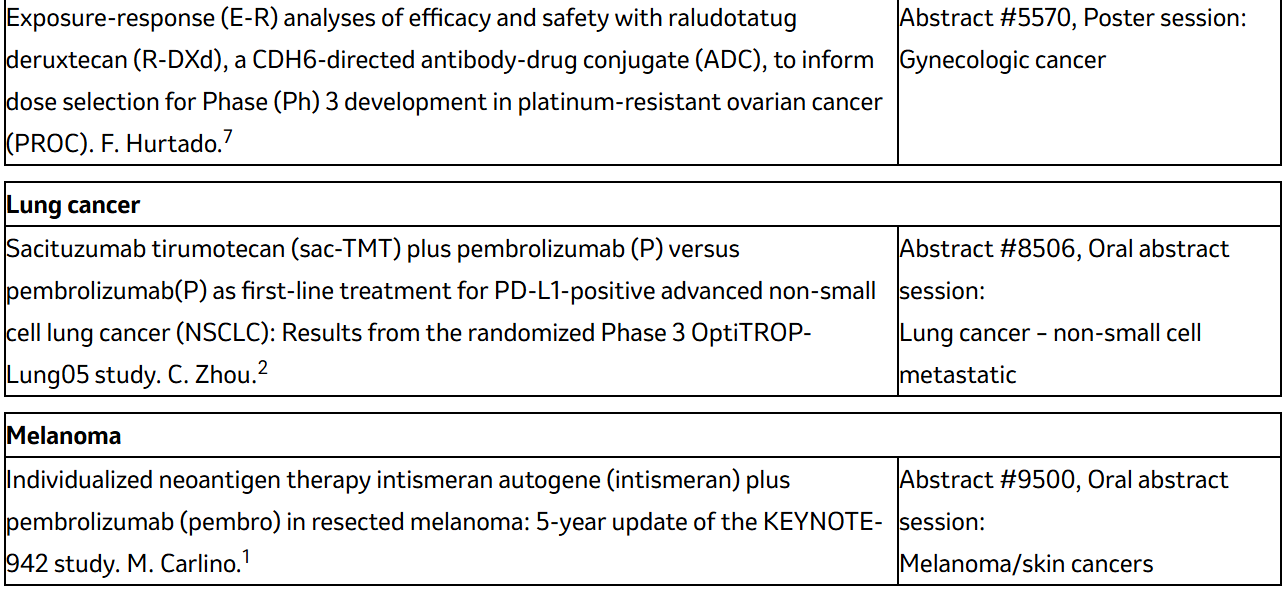
Details on abstracts listed above and additional key abstracts for Merck



About intismeran autogene (mRNA-4157 or V940)
Intismeran autogene is a novel investigational messenger RNA (mRNA)-based individualized neoantigen therapy (INT) consisting of a synthetic mRNA coding for up to 34 neoantigens that is designed and produced based on the unique mutational signature of the DNA sequence of the patient’s tumor. Upon administration into the body, the algorithmically derived and RNA-encoded neoantigen sequences are endogenously translated and undergo natural cellular antigen processing and presentation, a key step in adaptive immunity. Individualized neoantigen therapies are designed to train and activate an antitumor immune response by generating specific T-cell responses based on the unique mutational signature of a patient’s tumor.
About sacituzumab tirumotecan (sac-TMT)
Sac-TMT is an investigational TROP2-directed ADC with a belotecan-derived topoisomerase I inhibitor payload and a bifunctional linker designed with the potential to maximize payload delivery to tumor cells and minimize payload loss while circulating in the body. Sac-TMT is the only TROP2 ADC designed with a focus on both ends of the linker.
TROP2 is overexpressed on tumor cells compared to healthy cells in many common cancers, and through the TroFuse clinical development program, Merck is evaluating sac-TMT in 17 ongoing global Phase 3 trials across multiple tumor types, the broadest range of disease and treatment settings compared to any TROP2-directed ADC to date. The TroFuse development program spans early‑to-late‑stage disease in more than nine disease areas and includes more than 15,000 patients worldwide. Numerous Phase 3 trials are exploring sac-TMT as monotherapy and in combination with immunotherapies, aiming to improve survival and quality of life for patients with advanced and earlier-stage cancers.
About raludotatug deruxtecan (R-DXd)
Raludotatug deruxtecan is an investigational, potential first-in-class CDH6 directed ADC. Designed using Daiichi Sankyo’s proprietary DXd ADC Technology, raludotatug deruxtecan is comprised of a humanized anti-CDH6 IgG1 monoclonal antibody attached to a number of topoisomerase I inhibitor payloads (an exatecan derivative, DXd) via tetrapeptide-based cleavable linkers.
About KEYTRUDA® (pembrolizumab) injection for intravenous use, 100 mg
KEYTRUDA is an anti-programmed death receptor-1 (PD-1) therapy that works by increasing the ability of the body’s immune system to help detect and fight tumor cells. KEYTRUDA is a humanized monoclonal antibody that blocks the interaction between PD-1 and its ligands, PD-L1 and PD-L2, thereby activating T lymphocytes which may affect both tumor cells and healthy cells.
Merck has the industry’s largest immuno-oncology clinical research program. There are currently more than 2,800 trials studying KEYTRUDA across a wide variety of cancers and treatment settings. The KEYTRUDA clinical program seeks to understand the role of KEYTRUDA across cancers and the factors that may predict a patient's likelihood of benefitting from treatment with KEYTRUDA, including exploring several different biomarkers.
About the Merck and Kelun-Biotech strategic collaboration
Sac-TMT was developed by Kelun-Biotech. Kelun-Biotech (6990.HK) is a holding subsidiary of Kelun Pharmaceutical (002422.SZ), which focuses on the R&D, manufacturing, commercialization and global collaboration of innovative biological drugs and small molecule drugs. Under a collaboration agreement, Kelun-Biotech has granted Merck the exclusive rights to develop, manufacture and commercialize sac-TMT in all territories outside of Greater China (which includes Mainland China, Hong Kong, Macau and Taiwan).
About the Merck, Pfizer and Astellas collaboration
Merck previously entered a clinical collaboration agreement with Seagen and Astellas to evaluate the combination of Merck’s KEYTRUDA® (pembrolizumab) and Seagen’s and Astellas’ Padcev® (enfortumab vedotin-ejfv) in patients with muscle-invasive bladder cancer (MIBC) who are not eligible for or declined cisplatin-based chemotherapy. Padcev® and the Padcev device are trademarks jointly owned by Agensys, Inc., and Seagen Inc. Pfizer Inc. completed its acquisition of Seagen on December 14, 2023.
About the Daiichi Sankyo and Merck collaboration
Daiichi Sankyo and Merck entered into a global collaboration in October 2023 to jointly develop and commercialize ifinatamab deruxtecan (I-DXd), raludotatug deruxtecan (R-DXd) and patritumab deruxtecan (HER3-DXd), except in Japan where Daiichi Sankyo will maintain exclusive rights. Daiichi Sankyo will be solely responsible for manufacturing and supply. In August 2024, the global co-development and co-commercialization agreement was expanded to include gocatamig (MK-6070/DS3280), which the companies will jointly develop and commercialize worldwide, except in Japan where Merck will maintain exclusive rights. Merck will be solely responsible for manufacturing and supply for gocatamig.
Merck’s focus on cancer
Every day, we follow the science as we work to discover innovations that can help patients, no matter what stage of cancer they have. As a leading oncology company, we are pursuing research where scientific opportunity and medical need converge, underpinned by our diverse pipeline of more than 20 novel mechanisms. With one of the largest clinical development programs across more than 30 tumor types, we strive to advance breakthrough science that will shape the future of oncology. By addressing barriers to clinical trial participation, screening and treatment, we work with urgency to reduce disparities and help ensure patients have access to high-quality cancer care. Our unwavering commitment is what will bring us closer to our goal of bringing life to more patients with cancer.
About Merck
At Merck, known as MSD outside of the United States and Canada, we are unified around our purpose: We use the power of leading-edge science to save and improve lives around the world. For more than 130 years, we have brought hope to humanity through the development of important medicines and vaccines. We aspire to be the premier research-intensive biopharmaceutical company in the world – and today, we are at the forefront of research to deliver innovative health solutions that advance the prevention and treatment of diseases in people and animals. We foster a diverse and inclusive global workforce and operate responsibly every day to enable a safe, sustainable and healthy future for all people and communities.