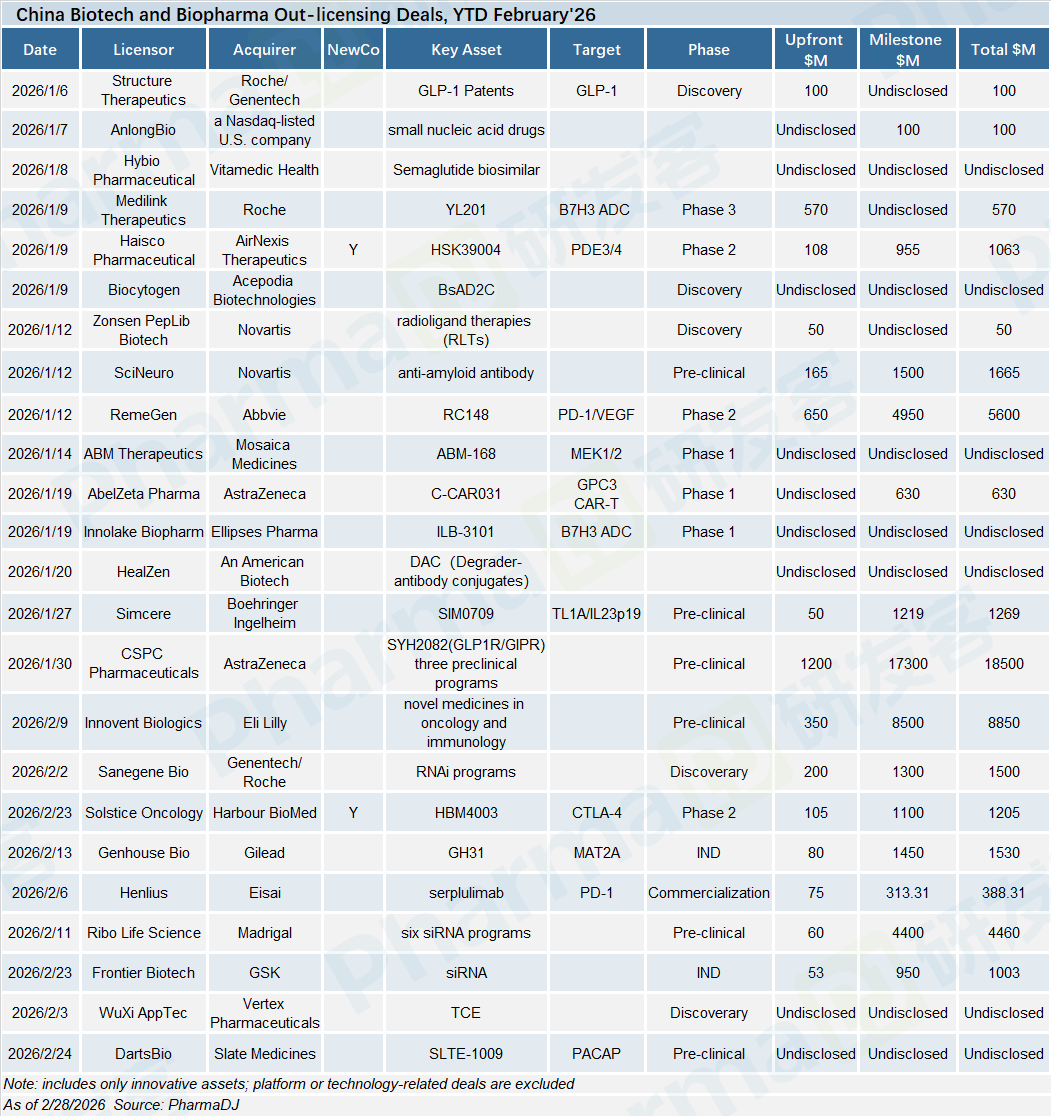
Chinese Biotech financing has been on a rebound since the second half of 2025, and this positive momentum has continued into the first two months of 2026.
A total of 42 financing deals were closed in January and February 2026, raising $1.36 billion — representing a 26% increase in deal count and a sharp 125% jump in financing volume compared with the same period last year.
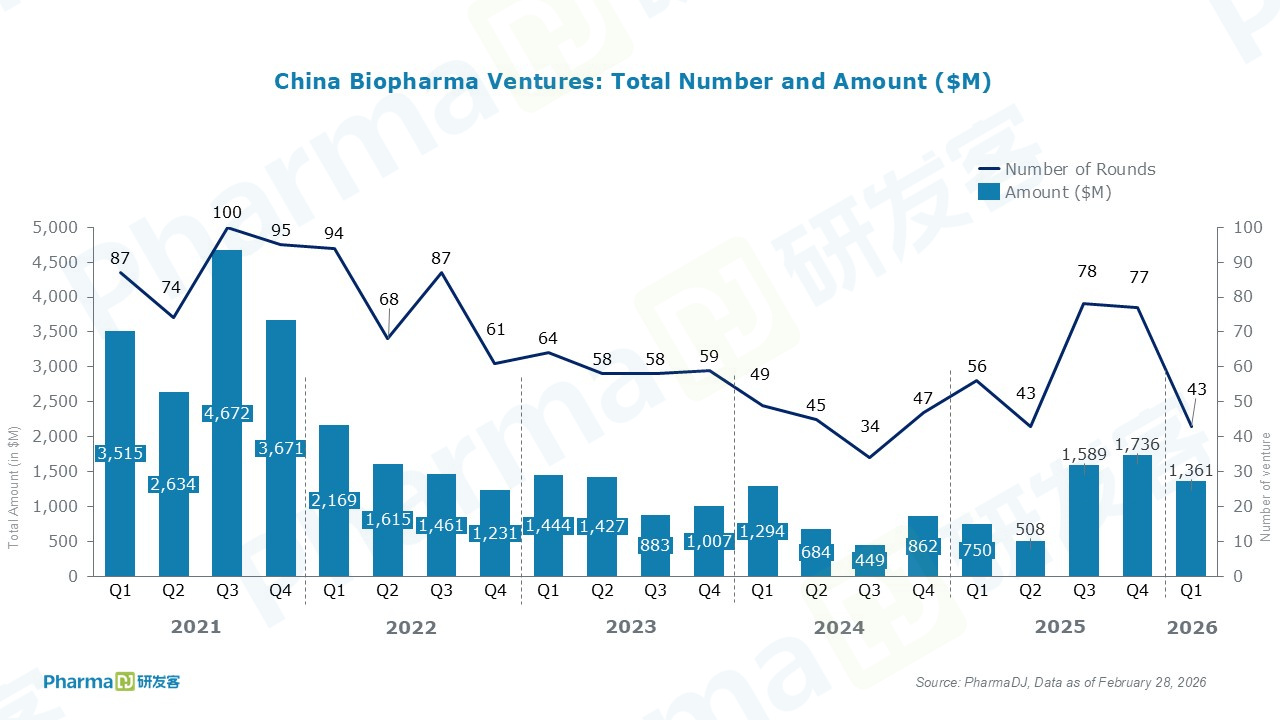
In February, Angitia Biopharmaceuticals secured the largest financing round of the period, completing a $130 million Series D financing. The round was co-led by Frazier Life Sciences and Venrock Healthcare Capital Partners. Proceeds will be used to advance clinical trials for three bone disease prospects: AGA111, AGA2115, and AGA2118.
Meanwhile, multinational pharmaceutical companies (MNCs) have increased their investments in high-quality Chinese biotech assets.
Lilly Asia Ventures (LAV) and Sanofi jointly led an oversubscribed $85 million Series B financing for QuantX Biosciences. The company expects to use the funding to advance its lead oral small molecule programs and support continued discovery efforts across its computational platform.
Separately, GluBio Pharmaceutical received a $30 million strategic equity investment from Sanofi. The capital will be deployed to advance two core programs — GLB-005 and GLB-007 — for the treatment of sickle cell disease.
In February, Chinese drugmakers recorded a total of 10 out licensing deals. Among them, Innovent Biologics struck its seventh collaboration with Eli Lilly. Innovent will receive an upfront payment of $350 million, plus up to approximately $8.5 billion in milestone payments.
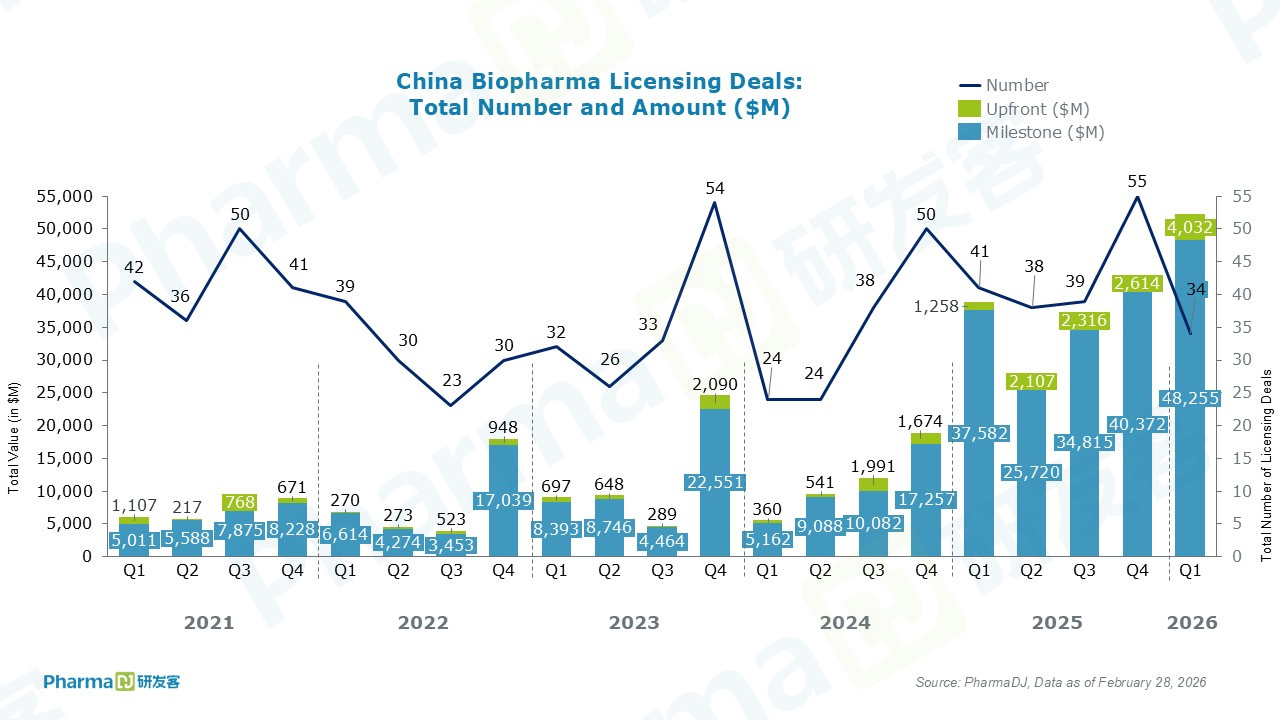
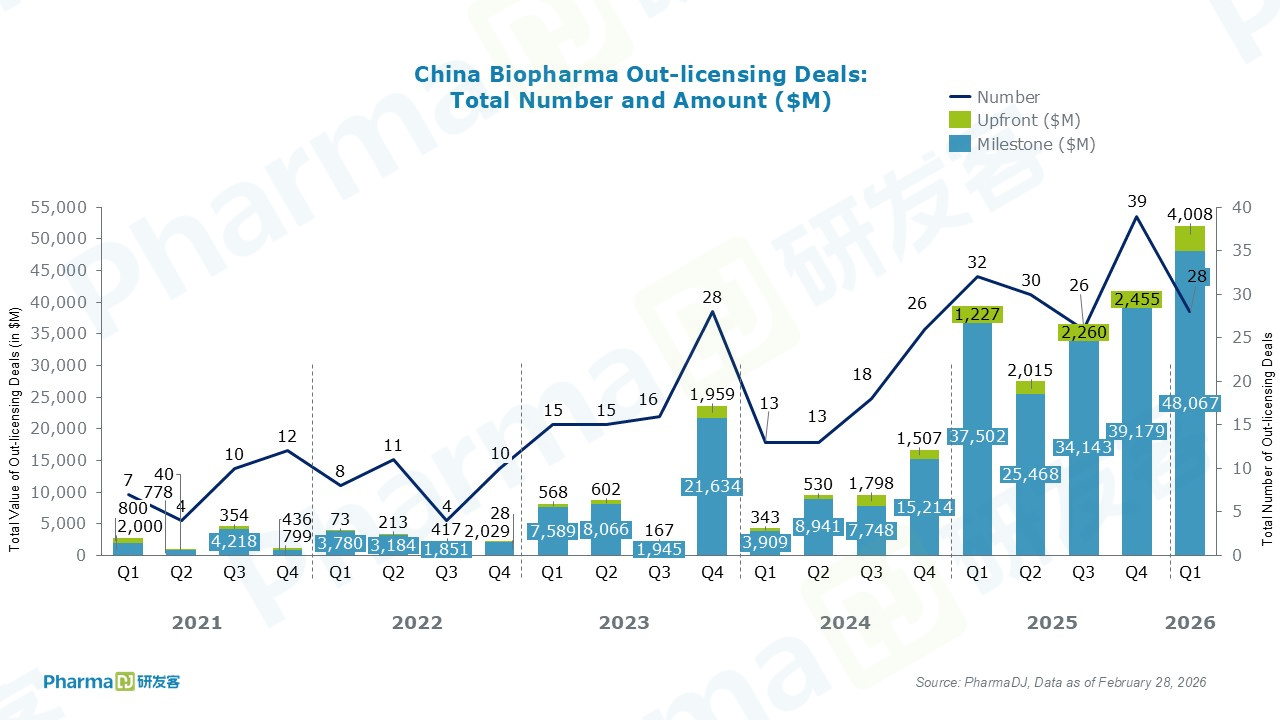
Chinese small interfering RNA (siRNA) programs gained notable momentum in February.
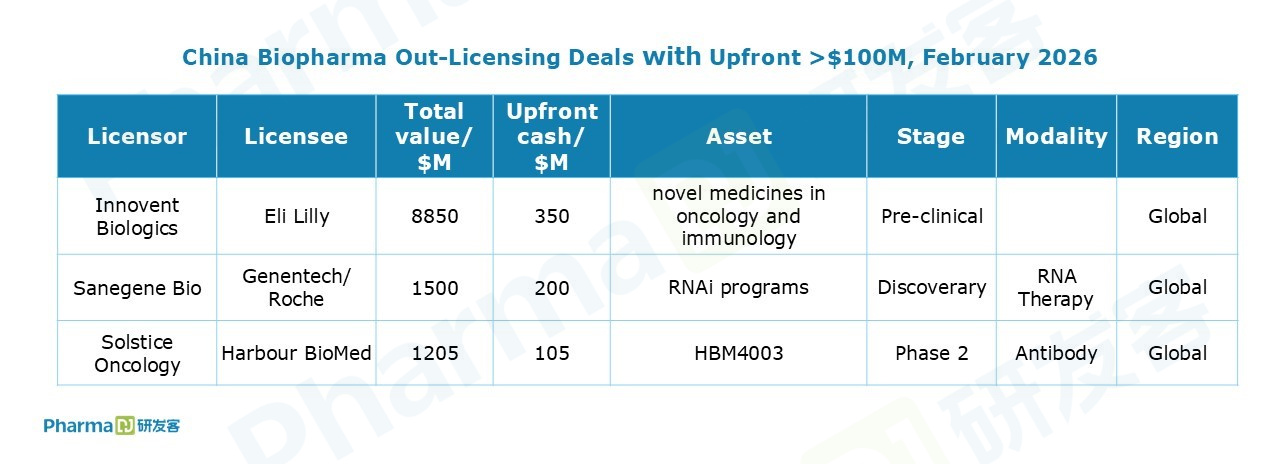
Sanegene Bio out-licensed an siRNA drug candidate to Genentech. Ribo Life Science entered a partnership with Madrigal to develop 6 preclinical siRNA therapeutics for the treatment of MASH. GSK paid $40 million upfront for a pair of oligonucleotide therapies from Frontier Biotech.