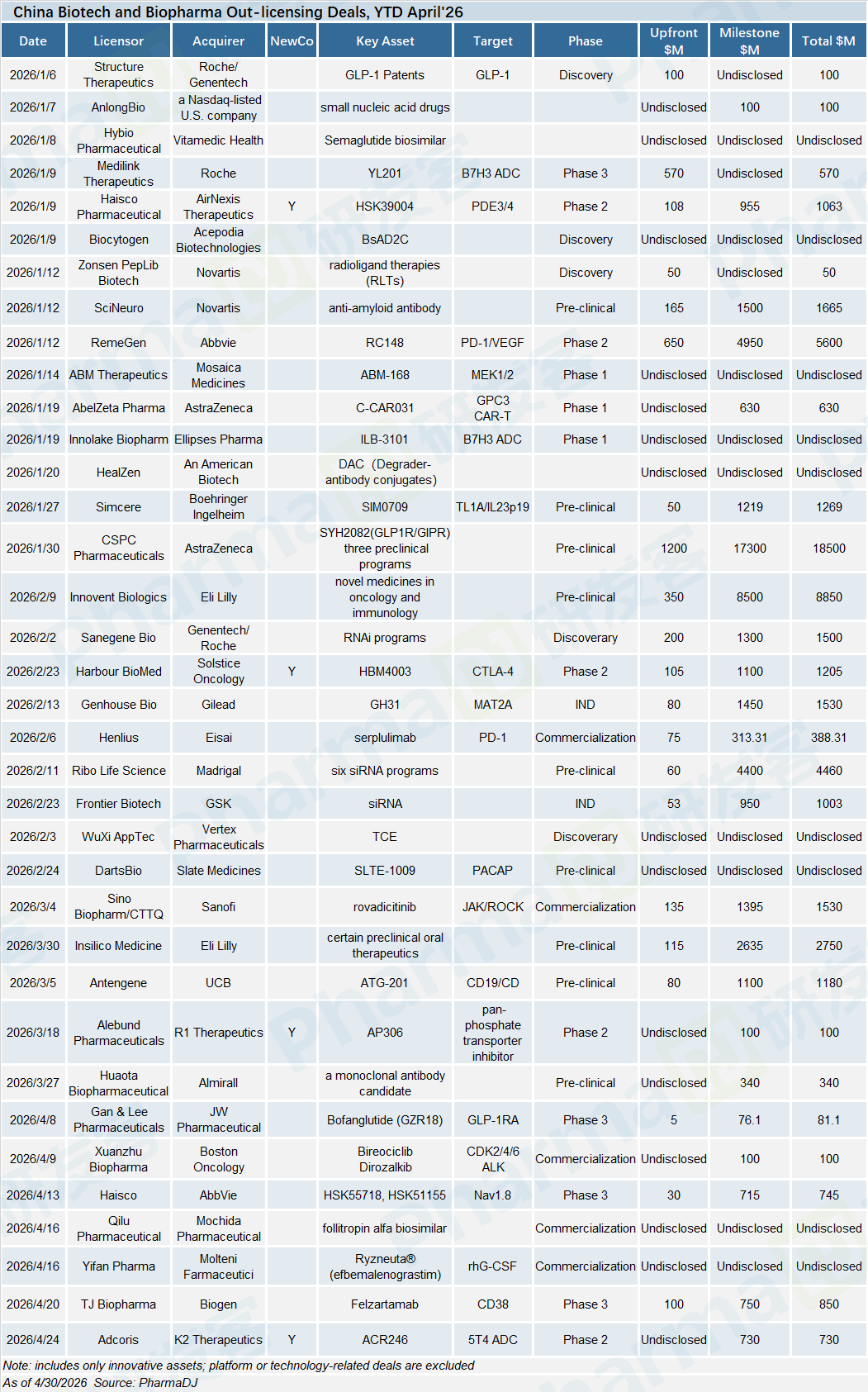
China’s biotech financing landscape cooled markedly in April, after a red-hot March deal spree. Chinese drugmakers closed 19 funding rounds totaling $574 million, compared with 30 rounds worth $1.59 billion recorded in March.
New Radiomedicine Technology(NRT) claimed April’s largest financing haul with a Series E round of over RMB 1 billion, setting a new all-time high for single-round fundraising in China’s radiopharmaceuticals space.
The global M&A boom for in vivo CAR-T therapies continued to stir Chinese funding. Oricell Therapeutics closed a $110 million Pre-IPO round in April. Meanwhile, VIVACTA Biotechnology, which develops in vivo CAR-T candidates leveraging both LNP and lentivirus delivery platforms, secured more than $50 million via its Series A and A+ financings.
QIMING Venture Partners kept doubling down on high-risk innovative drug R&D. In April, the VC firm led a $54 million Series A round for Vivatides Therapeutics, a developer focused on extrahepatic siRNA delivery technologies. QIMING also spearheaded nearly RMB 300 million Series B+ round for Novamab’s which specializes in innovative nanobody drug development.
On April 28, Mabwell Bioscience successfully listed on the Hong Kong Stock Exchange, raising approximately HK$1.189 billion (around RMB 1.035 billion). The proceeds will be primarily used to advance clinical development programs across oncology and age-related disease indications.
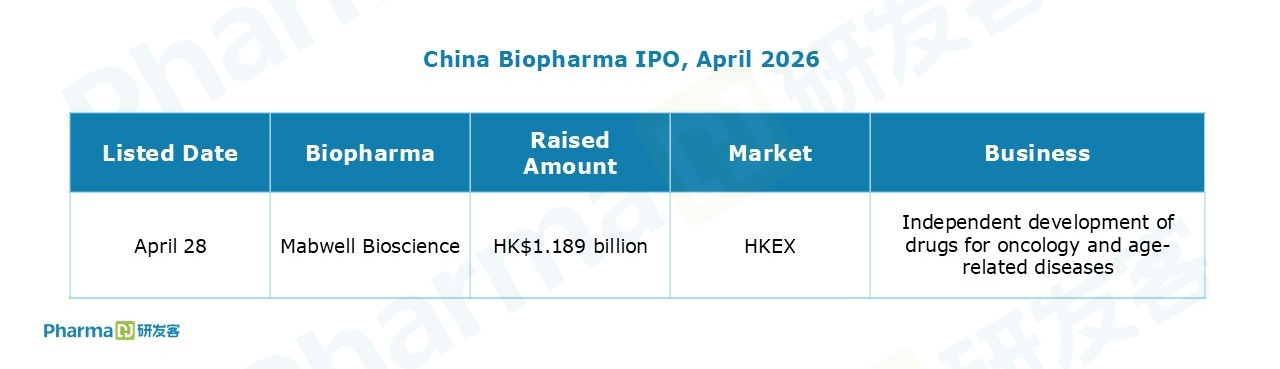
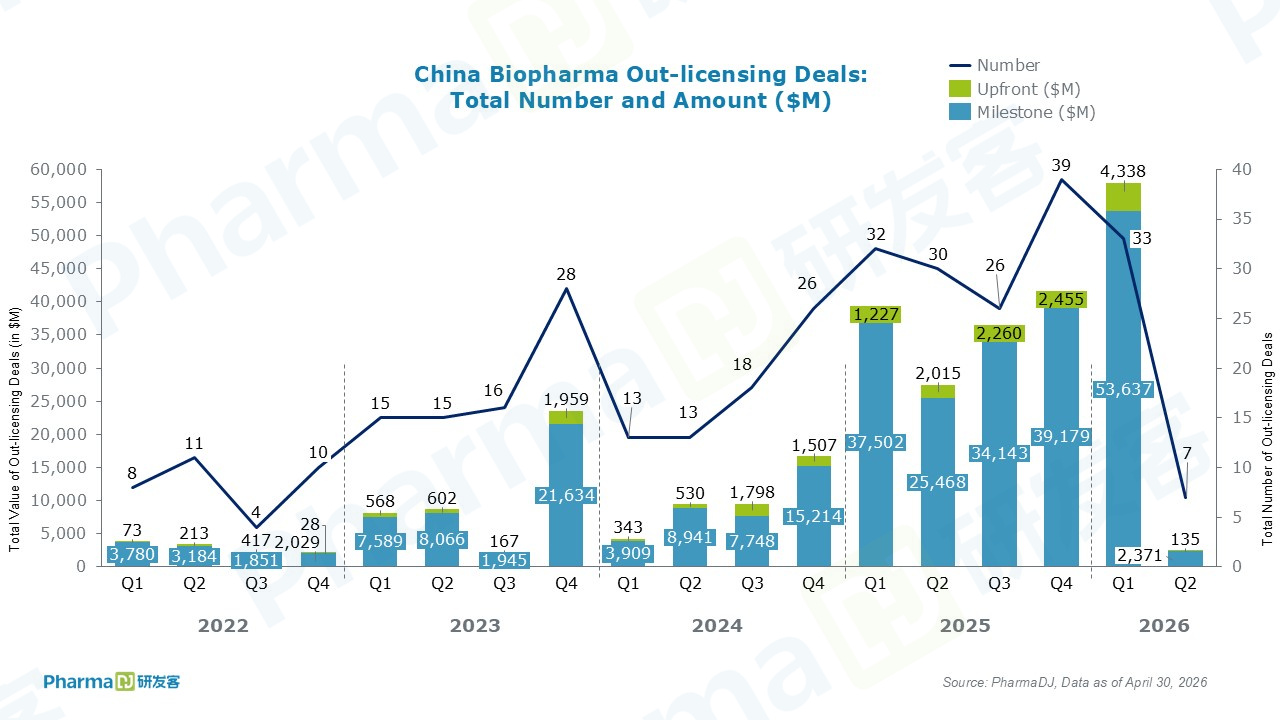
Chinese companies inked seven out-licensing deals in April.
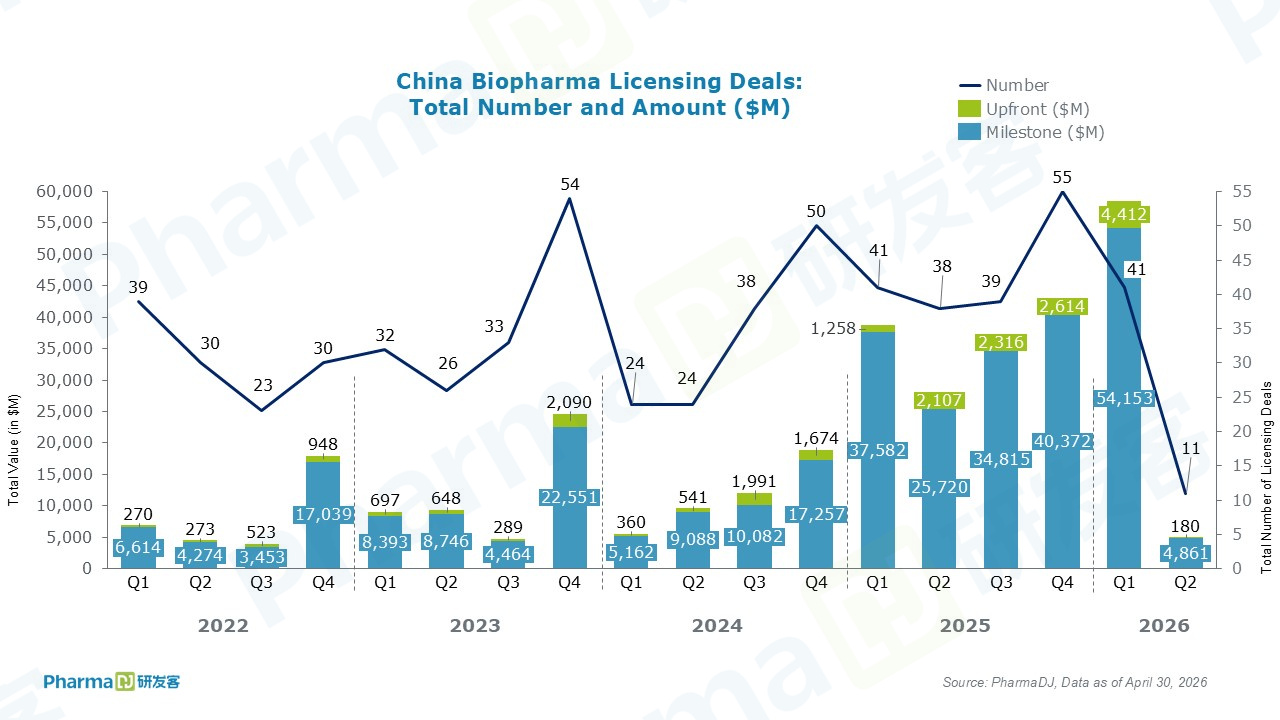
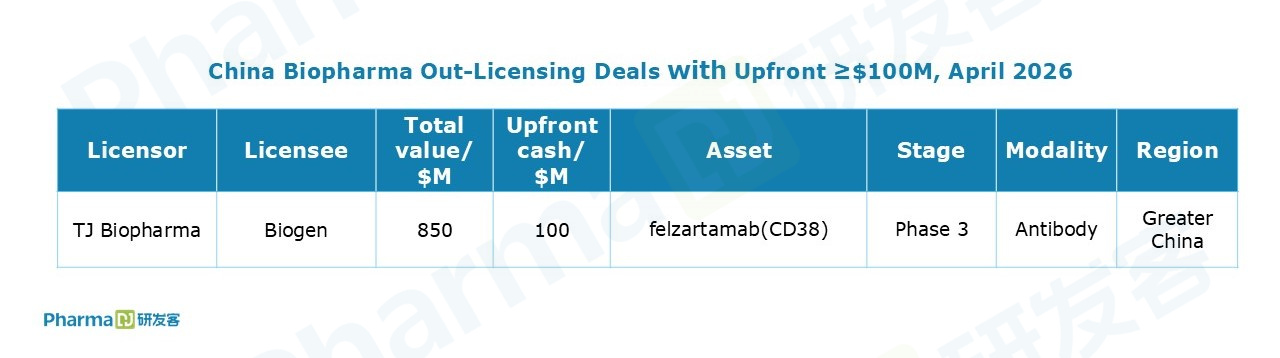
Notably, TJ Biopharma reached an agreement with Biogen to grant the latter exclusive rights to felzartamab in Greater China (covering Chinese Mainland, Hong Kong, Macau, and Taiwan). The deal entitles TJ Biopharma to a $100 million upfront payment and up to $750 million in potential milestone payments. Back in July 2024, Biogen had already obtained ex-Greater China rights to the CD38 monoclonal antibody through its acquisition of HI-Bio.
Separately, Haisco Pharmaceutical Group licensed two Nav1.8 inhibitor programs to AbbVie, netting a $30 million upfront plus up to $715 million in additional milestone payments.