Chunlin Xin, Vice President of External Collaboration at CanSino Biologics, told PharmaDJ that “using the adenovirus type 5 vector may be the reason why we succeeded with making the world first inhaled COVID-19 vaccine.”
CanSino’s Convidecia (AD5-nCOV), Source: CanSino Biologics
On October 26, Shanghai started to offer CanSino’s inhaled COVID-19 vaccine as a booster dose, making it the first city to offer the inhaled vaccine. This also happened to mark the official launch of the vaccine in China.
Just six days later, the company annouced that 13 cities of Jiangsu province were preparing to start using the inhaled vaccine. The news caused its stock to jump over 170% in Hong Kong and 69% in Shanghai in the past eight days, despite a generally gloomy stock market.
“The inhaled vaccine can induce respiratory mucosal immunity, like an invisible mask, to provide better protection for potential blocking infection,” Chunlin Xin, CanSino’s Vice President of External Collaboration, told PharmaDJ.

Xin Chunlin, CanSino’s Vice President of External Collaboration
The vaccine, marketed as Convideia Air, is a recombinant adenovirus type 5 vector (Ad-5) vaccine. It was initially approved for emergency use in adults aged 18 years or older by China’s drug adminstration agency in September 2022. It happens to be the first inhaled COVID-19 vaccine green lighted in the world.
“We did not do any formualtion adjustment, the inhalable version has the same formualtion as the intramuscular version, which list one of 11 WHO pre-qualified convid-19 vaccines for world wide use,” said Xin.
“We use a special nebulizing device from Aerogen to make the vaccine into small droplet like mist and inhale to the lung through the mouth. The entire vaccination process needs to be performed with the assistance of a trained healthcare worker.”

One Shanghai citizen received CanSino’s inhalaed vaccine, Source: Chinanews.com
28 days after booster immunization with Convideia Air, the neutralizing antibodies against the Omicron strain was 14 times that of an inactivated homologous booster and 6 times that of a recombinant protein vaccine, according to data released by CanSino. The protection lasts for at least 6 months.
“The dose of the inhalable version is only 20% of the intramuscular version. Therefore, the clincial results of over 10,000 adults showed that Covidecia Air’s overall adverse effects are lower, espeically with fever, and no injection site reactions”, Xin said. “However, we have not administered the vaccine to people with respiratory diseases yet”.
It is reported that the company has already started the adolescents trial aged 6-17 years.
The differences between CanSino and AstraZeneca inhalation vaccines
Just four weeks ago, AstraZeneca released the phase I study results of the intranasal formulation of its COVID-19 vaccine Vaxzevria. The data showed that the nasal spray failed to induce a consistent mucosal antibody response or strong systemic response.
“We used the human adenovirus type 5 as the vector to deliver the spike protein subunit of SARS-CoV-2”, Xin said. Adenovirus Type 5 is a very mild or no symptom virus which naturally infect human respiratory area.
“About 70% Chinese population has been infected by the adenovirus-5, and has the antibodies against Ad-5,” Xin explained to PharmaDJ. Therefore, they were not sure if these pre-existing antibodies would weaken the vector delivery efficacy at first.
Fortunately, CanSino found that the clinical results showed that the vaccine protect efficacy are very similar compare with other companies using different serum types of adenovirus.
AstraZeneca’s Vaxzevria, which also is a viral vector vaccine, uses a chimpanzee adenovirus (ChAdOx1) vector encoded with the SARS-CoV-2 spike protein gene.
Xin thinks that perhaps the delivery efficacy of ChAdOx1 is not good enough when inoculate by nasal spray. He suggested that “try using the inhalation like CanSino or to change the formulation, just as Bharat Biotech did.” The Indian company Bharat has successfully marketed its intranasal vaccine iNCOVACC, which also used chimpanzee adenovirus, but change its formulation to improve the delivery.
CanSino chose to explore administrating the vaccine to the lungs directly via the mouth, instead of the intranasal method.
“We tried both ways in the animal trials, and found that delivering it through mouth has better results,” Xin told PharmaDJ.
“We chose a Aerogen as our partner to develop the inhalation vaccine, because Aerogen is one of the best company which has more than 20 years innovative respiratory drug delivery experience,” said Xin, who added that the Aerogen-made nebulizer can aerosolize the vaccine to the droplets which the size is smaller than five micrometers to penetrate into deep lung area.
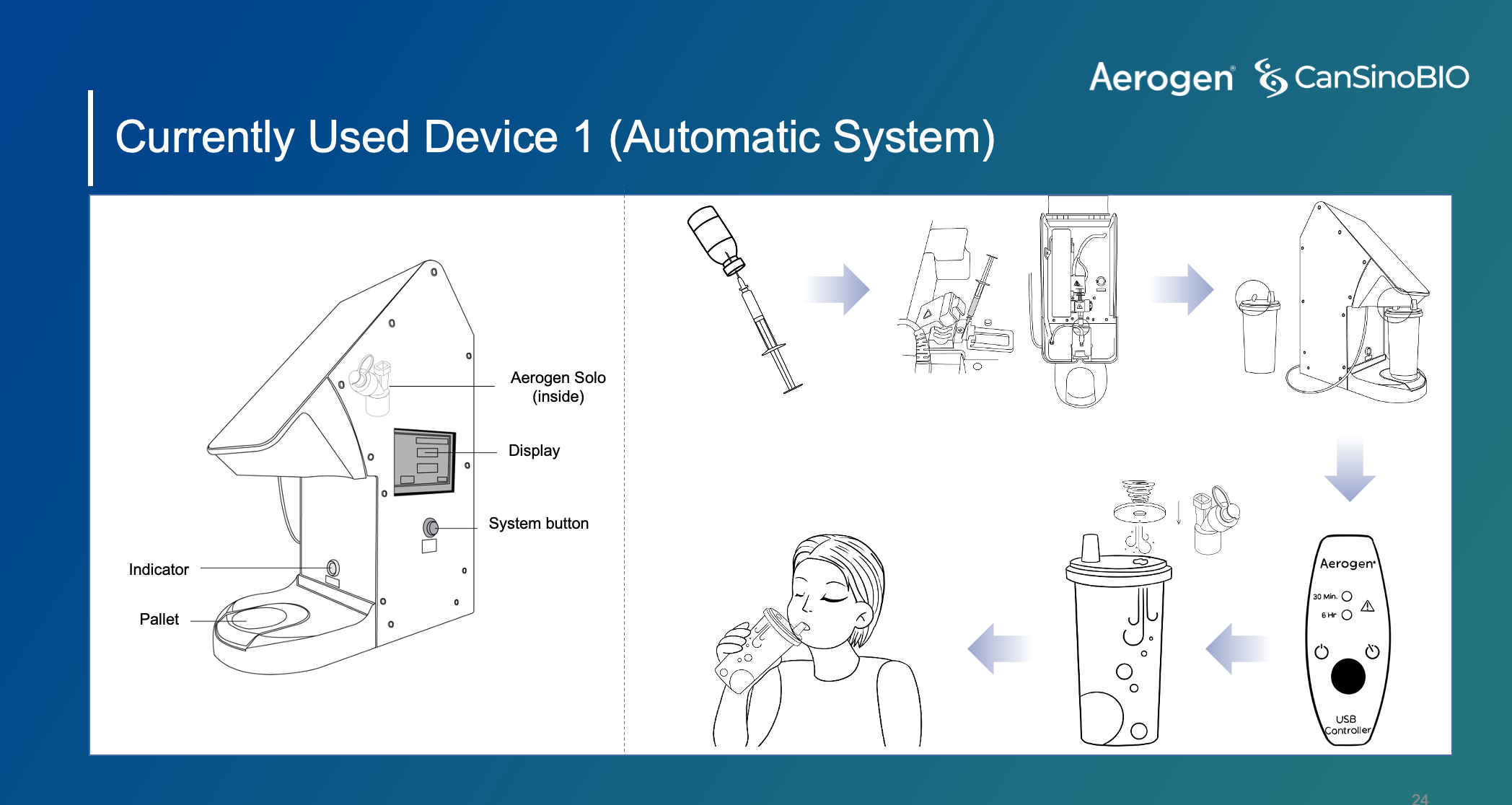
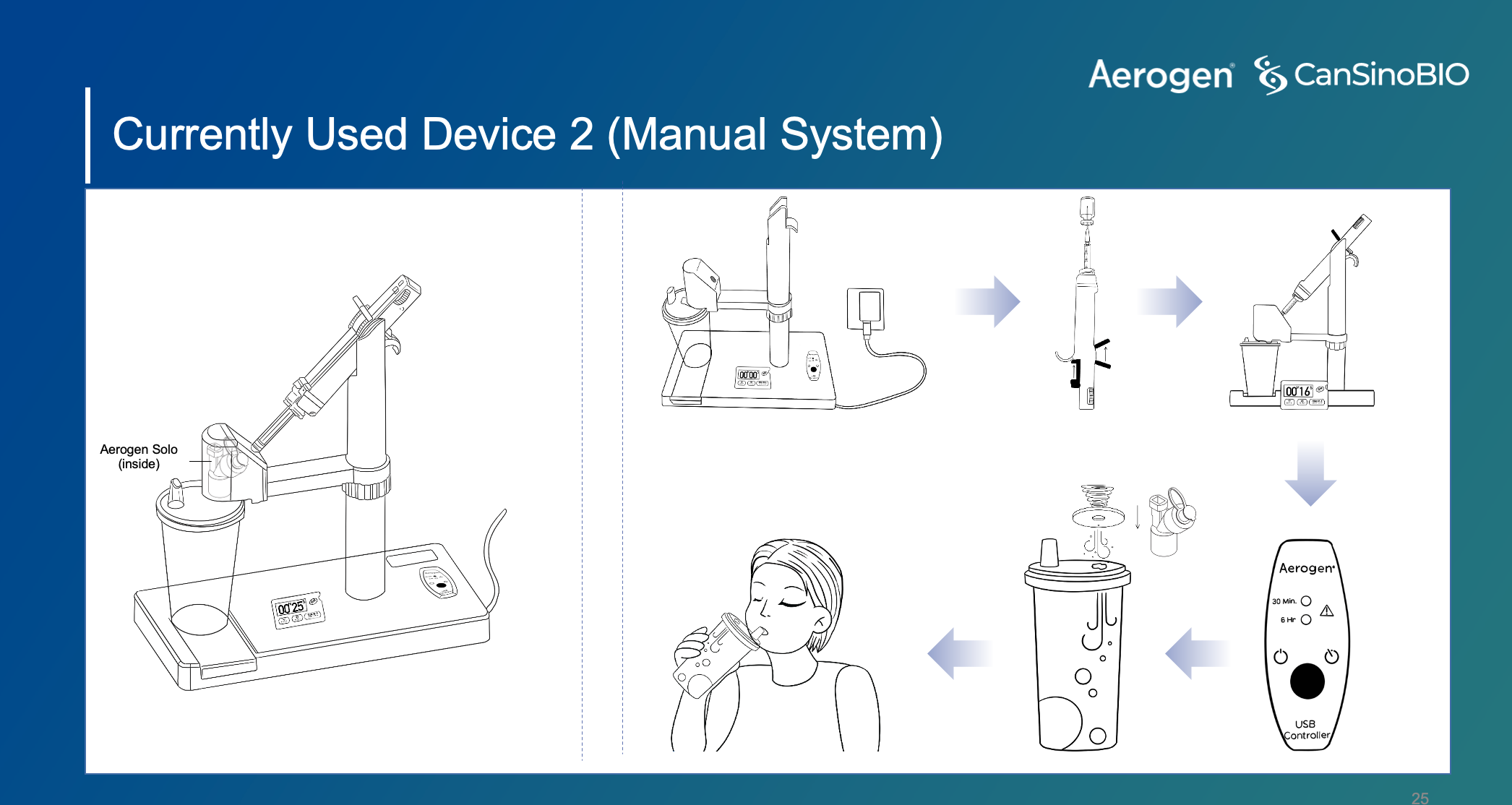
Nebulizer used in CanSino’s inhalaed vaccine, Source: CanSino Biologics
Xin also explained why CanSino only use 0.1ml of the inhaled vaccine. “The lungs are very sentive, so we only use 20% of the dosage of the intramuscular version to ensure the safety. The data has shown that the 0.1ml dosage induced the right immune response effectively.”
There are currently dozens of COVID-19 vaccines marketed in the world, with 11 included on the World Health Organization’s Emergency Use Listing. In China, nine have been approved. But CanSino continue to develop new vaccines, including Convideia Air, as it sees a need for it.
In Xin’s opinion, the existing vaccines, including the inactivated vaccines, offer good protection in preventing serious illness and death. But Omicron variant, the highly transmissible strain has emerged and produced more than 100 mutant variants in a short time.
“Therefore, it is still needed to improve the protection against more infections. I think that the development work of inhaled vaccines would be increasing in the future,” Xin said.
“This delivery method can potentially block the infection route of respiratory infectious diseases, and the inhaled vaccines can induce the local mucosal immune response and produce high affinity secreted IgA to stop the transmission by neutralizing the virus in entry air way, just like invisible masks.”
CanSino’s mRNA vaccine in phase II

CanSino’s pipeline, Source: CanSino Biologics
As a vaccine specialist, CanSino is also developing an mRNA vaccine for COVID-19.
Xin told PharmaDJ that they have already completed the phase I study and published the data. The company now is recruiting participants for the phase II study.
Since this year, the sales of mRNA vaccines have dropped quickly. But there has yet to be an mRNA COVID-19 vaccine approved in China.
“We started the development of the mRNA vaccine before the Omicron variants emerged,” Xin said. “Our new mRNA vaccine includes different sequence design potential not only target on current Omicron variant but also against future variant.”
(Edited by David Ho)